In the qualitative cation analysis procedure, Bi 3+ is detected by the appearance of a white precipitate
Question:
In the qualitative cation analysis procedure, Bi3+ is detected by the appearance of a white precipitate of bismuthyl hydroxide, BiOOH(s):
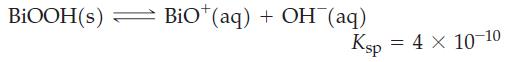
Calculate the pH of a saturated aqueous solution of BiOOH.
Transcribed Image Text:
BIOOH(s) BiO (aq) + OH(aq) Ksp = 4 X 10-10
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
To calculate the pH of a saturated solution of bismuthyl hydroxide BIOOH we can use the solubility p...View the full answer

Answered By

Danish Sohail
My objective is to become most reliable expert for clients. For last 10 years I have been associated with the field of accounting and finance. My aim is to strive for best results and pay particular attention to client needs. I am always enthusiastic to help clients for issues and concerns related to business studies. I can work on analysis of the financial statements, calculate different ratios and analysis of ratios. I can critically evaluate stock prices based on the financial analysis and valuation for companies using financial statements of the business entity being valued with use of excel tools. I have expertise to provide effective and reliable help for projects in corporate finance, equity investments, financial accounting, cost accounting, financial planning, business plans, marketing plans, performance measurement, budgeting, economic research, risk assessment, risk management, derivatives, fixed income investments, taxation, auditing, and financial performance analysis.
4.80+
78+ Reviews
112+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
A sample containing an alkali sulfate is dried, weighed and dissolved in dilute HCl. Barium chloride solution is added in excess to precipitate barium sulfate, and the precipitate is digested in the...
-
The composition of rewards applied by an organization and its association with performance management system can shape organizational culture, Do you agree or disagree and why?
-
1.) Let G be an n-partite simple graph, the partite sets of which are {ui, vi}, {u2, v2}, {un, Un}. Assume that the sequence of degrees d(v), d(u2), d(v2),..., d(un-1), d(vn-1), d(un), d(vn) is some...
-
FinCorp Inc. is exploring different portfolio allocations between two stocks. Complete the following table. Case1 Case 2 Case 3 Case 4 Case 5 $ invested in stock 1 $ invested in stock 2 Total $...
-
Distinguish between taxable temporary differences and deductible temporary differences, and give at least two examples of each type.
-
You began the systems analysis phase at New Century Health Clinic by completing a series of interviews, reviewing existing reports, and observing office operations. (Your instructor may provide you...
-
Abbott Industries is a well-known supplier of pharmaceuticals worldwide. Founded by Dr. Wallace Abbott, the company was incorporated in 1900 after he had been developing and making pharmaceuticals...
-
Capital budgeting methods, no income taxes. City Hospital, a non-profit organization, estimates that it can save $28,000 a year in cash operating costs for the next 10 years if it buys a...
-
1. You invest $2000 in an account that pays simple interest of 7% for 10 years. The amount of money you'll have at the end of 10 years is: 2. Use the compound interest formula to determine the...
-
A solution is saturated with magnesium palmitate [Mg(C 16 H 31 O 2 ) 2 , a component of bathtub ring] at 50 C. How many milligrams of magnesium palmitate will precipitate from 965 mL of this solution...
-
Fluoridated drinking water contains about 1 part per million (ppm) of F. Is CaF 2 sufficiently soluble in water to be used as the source of fluoride ion for the fluoridation of drinking water?...
-
Why is the organ demand curve downward-sloping rather than vertical?
-
Question Content AreaIf merchandise that cost the company $17,000 is expected to be returned in the coming year, the required adjustment _____. a. increases Estimated Returns Inventory and decreases...
-
The goal is to reduce wastefulness and develop a tight, efficient budget. The management team knows that this will take time, so they plan to allow more time and additional resources in the budget...
-
Cormac purchased furniture from a local business with no payments for 12 months at 0% interest. Six months later, he was told he owed the original amount, plus interest and late fees. When he finally...
-
Plot the number 0.0015 on the log scale below. 105 104 103 102 101 100 101 102 103 104 105
-
Question 46 (15 points) Complete the following java code as stated below: import java.util.Scanner; public class Example { //----- //This program reads the amount of money spent by customers and...
-
Rockwell Corporation uses a periodic inventory system and has used the FIFO cost method since inception of the company in 1976. In 2011, the company decided to switch to the average cost method. Data...
-
You've been asked to take over leadership of a group of paralegals that once had a reputation for being a tight-knit, supportive team, but you quickly figure out that this team is in danger of...
-
(a) Jenny Kent asks your help in understanding the term activity index. Explain the meaning and importance of this term for Jenny. (b) State the two ways that variable costs may be defined.
-
A. J. Hernandez claims that the relevant range concept is important only for variable costs. (a) Explain the relevant range concept. (b) Do you agree with A. J.s claim? Explain.
-
The relevant range is indispensable in cost behavior analysis. Is this true? Why or why not?
-
3. You are cutting a steel rod with a cutting velocity of 75. m/min and notice that your tool wears out after 330 minutes. You switch to a cutting velocity of 150 m/min (keeping the same cooling...
-
2. In a turning process, you are using a tool with a rake angle of 30. You measure the shear angle to be 45. Draw the Merchant's circle. Label all 7 forces, all angles, and include the tool tip in...
-
An incompressible, steady, viscous, and fully developed flow is established between two horizontal parallel plates by moving the upper plate with a constant velocity of U in the (+) X direction,...

Study smarter with the SolutionInn App


