Pressure cookers achieve a high cooking temperature to speed the cooking process by heating a small amount
Question:
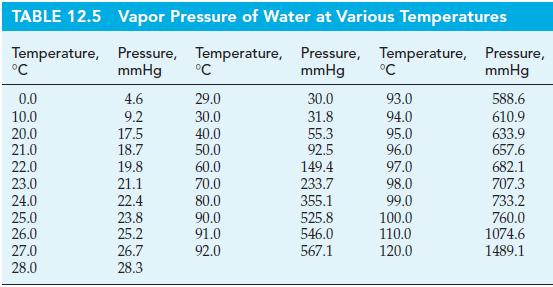
Pressure cookers achieve a high cooking temperature to speed the cooking process by heating a small amount of water under a constant pressure. If the pressure is set at 2 atm, what is the boiling point of the water? Use information from Table 12.5.
Table 12.5
Transcribed Image Text:
TABLE 12.5 Temperature, °C 0.0 10.0 20.0 21.0 22.0 23.0 24.0 25.0 26.0 27.0 28.0 Vapor Pressure of Water at Various Temperatures Pressure, Temperature, Pressure, Temperature, mmHg mmHg 4.6 9.2 17.5 18.7 19.8 21.1 22.4 23.8 25.2 26.7 28.3 °C 29.0 30.0 40.0 50.0 60.0 70.0 80.0 90.0 91.0 92.0 30.0 31.8 55.3 92.5 149.4 233.7 355.1 525.8 546.0 567.1 °C 93.0 94.0 95.0 96.0 97.0 98.0 99.0 100.0 110.0 120.0 Pressure, mmHg 588.6 610.9 633.9 657.6 682.1 707.3 733.2 760.0 1074.6 1489.1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
Temperature C Pressure mmHg 290 00 100 46 200 92 210 175 ...View the full answer

Answered By

Muhammad Ghyas Asif
It is my obligation to present efficient services to my clients by providing a work of quality, unique, competent and relevant. I hope you have confidence in me and assign me the order and i promise to follow all the instructions and keep time.
4.60+
109+ Reviews
203+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
What is the average return of a portfolio that has 10 invested in stock A 40 invested in stock B and 50 invested in stock C The return on stock A 13 8 return on stock B 12 6 and return on stock C 5
-
A sample containing an alkali sulfate is dried, weighed and dissolved in dilute HCl. Barium chloride solution is added in excess to precipitate barium sulfate, and the precipitate is digested in the...
-
A small amount of water and its saturated vapour are en- closed in a vessel at a temperature t = 100 C. How much (in per cent) will the mass of the saturated vapour increase if the temperature of the...
-
Describe how you plan to allocate your budget to each promotional activity. Provide a brief rationale. First, when developing a marketing budget plan, you want to start with the total budget...
-
A route salesperson for Ideal Milk Company delivered a onehalf- gallon glass jug of milk to Allens home. The next day, when Allen grasped the milk container by its neck to take it out of his...
-
Show the products obtained from addition of methyl magnesium bromide to the following compounds: (a) Cyclopentanone (b) Benzophenone (diphenyl ketone) (c) 3-Hexanone
-
What is the primary purpose of admission-seeking questions?
-
Using an Aging Schedule to Account for Bad Debts Carter Company sells on credit with terms of n/30. For the $500,000 of accounts at the end of the year that are not overdue, there is a 90%...
-
A clinical resear would like to know if his diet program is effective in lowering patients blood pressure. The following patient. Blood pressure before diet blood pressure after diet 1. 15. 12 2. 15....
-
One popular demonstration in chemistry labs is performed by boiling a small quantity of water in a metal can (such as a used soda can), picking up the can with tongs and quickly submerging it upside...
-
A double boiler is used when a careful control of temperature is required in cooking. Water is boiled in an outside container to produce steam, and the steam condenses on the outside walls of an...
-
This March 31, 2019, adjusted trial balance for RJs Baseball Academy has been alphabetized as follows. Prepare the closing entries and the post-closing trial balance for the company Account Name Dr....
-
Suppose you doubled the charge at some position in space. What would happen to its electric potential energy (assuming that it was not zero to start with)?
-
My question is what the following mean in a communication plan in project management. in this scenario I am a project manager making a communication plan while monitoring a project. What do I fill in...
-
Project Communication Management and Project Stakeholder Engagement. 9. Based on your experience and observations, list 5 of the most inefficient communications strategy that you have experienced so...
-
Create a communication escalation table for a project. What is the meaning of the following columns when it comes to communication escalation in project management? Priority Definition Decision...
-
Alpha Investment Services (AIS) Crisis Communication It's a generalized question. Answer and organize it in the same order. A-11.1 Identify the major controls in place and rate as Excellent, Good,...
-
Manufacturers Southern leased high-tech electronic equipment from International Machines on January 1, 2013. International Machines manufactured the equipment at a cost of $200,000. The equipment has...
-
To balance the chemical equation SiH3 + O2 SiO2 + HO, you could introduce coefficients a, b, c, d and write aSiH3 + bO2 cSiO + dHO then write linear equations for each element. The equation for Si...
-
Stock Valuation Suppose you know that a companys stock currently sells for $60 per share and the required return on the stock is 12 percent. You also know that the total return on the stock is evenly...
-
Stock Valuation No More Corp. pays a constant $11 dividend on its stock. The company will maintain this dividend for the next eight years and will then cease paying dividends forever. If the required...
-
Valuing Preferred Stock Ayden, Inc., has an issue of preferred stock outstanding that pays a $6.50 dividend every year in perpetuity If this issue currently sells for SI 13 per share, what is the...
-
Melinda invests $450,000 in a City of Heflin bond that pays 6 percent interest. Alternatively, Melinda could have invested the $450,000 in a bond recently issued by Surething Incorporated that pays...
-
Pacific Ink had beginning work-in-process inventory of $890,960 on October 1. Of this amount, $358,200 was the cost of direct materials and $532,760 was the cost of conversion. The 62,000 units in...
-
7) Compute the taxpayer's taxable income in each of the following cases: a) Jack is married and files a joint return with his wife Alice. They are both under age 65. Jack and Alice have two dependent...

Study smarter with the SolutionInn App


