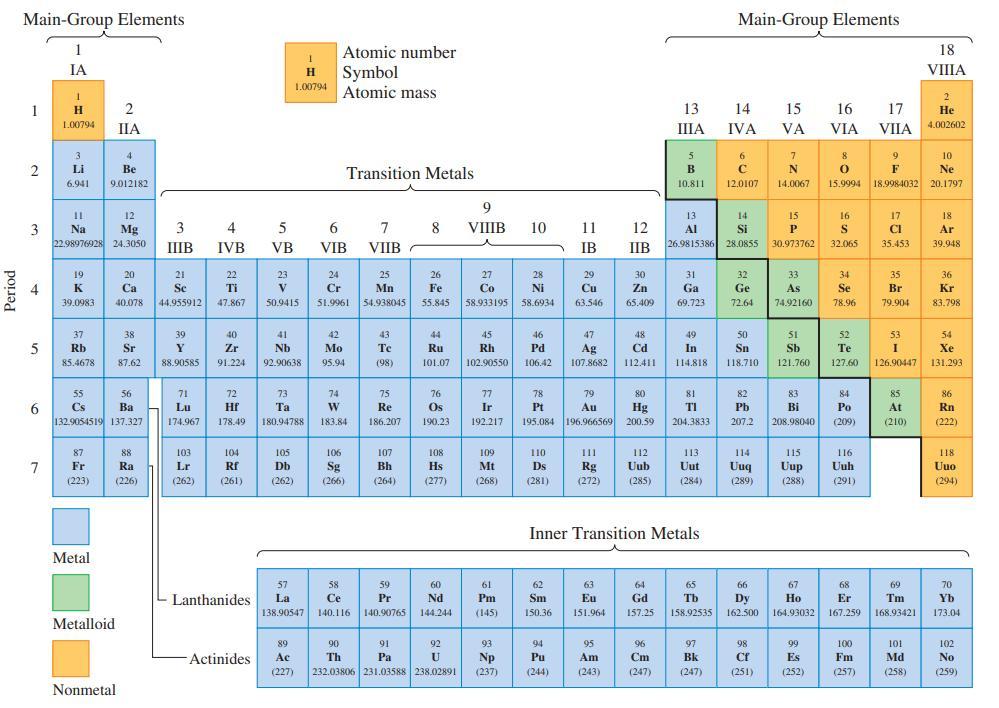
Refer only to the periodic table on the inside front cover and indicate which of the atoms,
Question:
Refer only to the periodic table on the inside front cover and indicate which of the atoms, Bi, S, Ba, As, and Ca,
(a) Is most metallic;
(b) Is most nonmetallic;
(c) Has the intermediate value when the five are arranged in order of increasing first ionization energy.
Transcribed Image Text:
Period Main-Group Elements 1 2 4 5 11 3 Na 6 IA 7 1 H 1.00794 3 Li 6.941 19 K 39.0983 12 Mg 22.98976928 24.3050 37 Rb 85.4678 55 Cs 87 Fr (223) 4 Be 9.012182 Metal 2 IIA 56 Ba 132.9054519 137.327 Metalloid 20 Ca 40.078 38 Sr 87.62 Nonmetal 88 Ra (226) 3 IIIB 21 Se 44.955912 39 Y 88.90585 71 Lu 174.967 103 Lr (262) 4 IVB 22 Ti 47.867 40 Zr 91.224 72 Hf 178.49 104 Rf (261) Lanthanides Actinides 5 VB 23 V 50.9415 1 H Symbol 1.00794 Atomic mass 41 Nb 92.90638 73 Ta 180,94788 105 Db (262) Atomic number 6 VIB Transition Metals 24 Cr 51.9961 42 Mo 95.94 74 W 183.84 106 Sg (266) 57 58 La Ce 138.90547 140.116 7 8 VIIB 25 Mn 54.938045 43 Te (98) 75 Re 186.207 107 Bh (264) 59 Pr 140.90765 26 Fe 55.845 91 Pa 44 Ru 101.07 76 Os 190.23 108 Hs (277) 60 Nd 144.244 92 89 Ac (227) 232.03806 231.03588 238.02891 90 Th U 9 VIIIB 27 Co 58.933195 45 Rh 102.90550 77 Ir 192.217 109 Mt (268) 61 Pm (145) 93 Np (237) 10 28 Ni 58.6934 46 Pd 106.42 110 Ds (281) 11 12 IB IIB 78 79 Pt Au 195.084 196.966569 62 Sm 150.36 29 Cu 63.546 94 Pu (244) 47 Ag 107.8682 30 Zn 65.409 63 Eu 151.964 48 Cd 112.411 111 112 Rg Uub (272) (285) 95 Am (243) 80 Hg 200.59 64 Gd 157.25 13 IIIA 96 Cm (247) 5 B 10.811 Inner Transition Metals 31 Ga 69.723 49 In 114.818 13 14 Al Si 26.9815386 28.0855 81 TI 204.3833 113 Uut (284) 65 Tb 158.92535 Main-Group Elements 97 Bk (247) 14 IVA 6 с 12.0107 32 Ge 72.64 50 Sn 118.710 82 Pb 207.2 114 Uuq (289) 66 Dy 162.500 98 Cf (251) 15 VA 7 N 14.0067 15 P 30.973762 33 As 74.92160 51 Sb 121.760 83 Bi 208.98040 115 Uup (288) 67 Ho 164.93032 99 Es (252) 16 VIA 8 9 0 F 15.9994 18.9984032 16 S 32.065 34 Se 78.96 52 Te 127.60 84 Po (209) 116 Uuh (291) 68 Er 167.259 17 VIIA 100 Fm (257) 17 CI 35.453 35 Br 79.904 53 I 126.90447 85 At (210) 69 Tm 168.93421 101 Md (258) 18 VIIIA 2 He 4.002602 10 Ne 20.1797 18 Ar 39.948 36 Kr 83.798 54 Xe 131.293 86 Rn (222) 118 Uuo (294) 70 Yb 173.04 102 No (259)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a Most Metallic Metals are typically found on the left side of the periodic table especially in the ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Refer only to the periodic table on the inside front cover, and arrange the following ionization energies in order of increasing value: the first ionization energy of F; the second ionization energy...
-
(A) Refer only to the periodic table on the inside front cover, and arrange the following species in order of increasing size: Ti 2+ , V 3+ , Ca 2+ , Br - , and Sr 2+ . (B) Refer only to the periodic...
-
(A) Refer to the periodic table on the inside front cover, and arrange the following in the expected order of increasing first ionization energy Cl, K, Mg, S. (B) Refer to the periodic table on the...
-
31. Which combination of the compounds and their geometry are correct h) CIF, - V-shaped e) CIF, - Tshaped OB. a HeCl, - linoar 24. Which of the following havea bond angle smaller than tetrahodral...
-
How does the theory of comparative advantage relate to the currency swap market?
-
Use the direct method to determine cash flows from operating activities The following accounts and corresponding balances were drawn from Widjaja Company's 2012 and 2011 y ear -end balance sheets....
-
I spent time with you. It seems that this deed is out of character for you. You were not awarded your normal annual bonus. I would probably feel the same. Is that what happened? You normally wouldnt...
-
Church Company completes these transactions and events during March of the current year (terms for all its credit sales are 2/10, n/30). Mar. 1 Purchased $43,600 of merchandise from Van Industries,...
-
Fort Gaines guards the western entrance to Mobile Bay. In order to prevent enemy ships from passing, it needs to be able to cover about 1 km of the bay with cannon fire. The cannons fire projectiles...
-
Refer to the periodic table on the inside front cover and indicate (a) The most nonmetallic element; (b) The transition metal with lowest atomic number; (c) A metalloid whose atomic number is exactly...
-
Explain why the first ionization energy of Mg is greater that of Na, whereas the second ionization of Na is greater than that of Mg.
-
Repeat Problem 12.6.1 for the analysis of variance table shown in Figure 12.37.
-
Smith was represented by Lewis, an attorney, in a divorce action against her husband, General Smith. General Smith was employed by the California National Guard and, as a state employee, belonged to...
-
Answer the following questions, which require you to think critically about the legal principles that you learned in this chapter. Tameny had worked for Atlantic Richfi eld Company (ARCO) for 15...
-
During construction at Alma College, an inadequately reinforced wall of an excavation caved in and killed Clark, a worker employed by Beard, the general contractor. The contract between the...
-
Mansfi eld contacted his insurance agent and asked him to obtain an automobile insurance liability policy to be effective as of February 10. On February 14, Mansfi eld was involved in an accident...
-
Assume that the indirect quote is for 115 Japanese yen per U.S. dollar and that the direct quote is for 1.25 U.S. dollars per euro. What is the yen per euro exchange rate?
-
Record transactions and adjustments. Prepare an answer sheet with the column headings shown after the following list of transactions. Record the effect, if any, of the transaction entry or adjusting...
-
If (x) 0 on the interval [a, b], the definite integral gives the exact area under the curve between x = a and x = b.
-
Why arent transactions involving accounts payable considered to be financing activities?
-
Assume that a company repays a $300,000 loan from its bank and then later in the same year borrows $500,000. What amount(s) would appear on the statement of cash flows?
-
How do the direct and the indirect methods differ in their approach to computing the net cash provided by operating activities?
-
The items making up the statement of financial position of Phillips Truck Rental at December 31 are listed below in tabular form. Assets Liabilities Equity Balances $13,900 Cash + Accounts Receivable...
-
1. You are given two areas connected by a tie-line with the following characteristics Area 1 R=0.005 pu D=0.6 pu Base MVA = 500 Area 2 R=0.01 pu D=1.0 pu Base MVA = 500 A load change of 150 MW occurs...
-
2. (a) A two-area power system connected by a tie-line has the following characteristics on a common 1000 MVA base: Area 1 2 R R = 4% R2=5% D D = 0.5 D2=0.8 Each area has a generating unit and these...

Study smarter with the SolutionInn App


