Refer to Figure 19-23. Predict the electrode reactions and the overall reaction when the anode is made
Question:
Refer to Figure 19-23. Predict the electrode reactions and the overall reaction when the anode is made of
(a) Copper and
(b) Platinum.
Figure 19-23
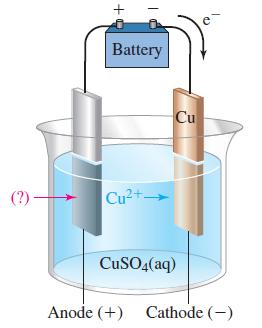
Transcribed Image Text:
(?)- Battery cass Cu2+ Cu CuSO4(aq) Anode (+) Cathode (-)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Analyze In both cases we have to decide on the likely oxidation and reduction processes The low reduction potential of Cu 2 aq makes this the likely r...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
A wire of length L = 4 ft and diameter d = 0.125 in. is stretched by tensile forces P = 600 lb. The wire is made of a copper alloy having a stress-strain relationship that may be described...
-
A circular bar of length L = 32 in. and diameter d = 0.75 in. is subjected to tension by forces P (see figure). The wire is made of a copper alloy having the following hyperbolic stress-strain...
-
Solve the linear equation with the x-intercept method. Check your answer. Approximate the solution to the nearest thousandth whenever appropriate. 2x = -(3x)
-
Tattle Company engaged in the following transactions in October 2010: Oct. 7 Sold merchandise on credit to Lina Ortiz, terms n/30, FOB shipping point, $6,000 (cost, $3,600). 8 Purchased merchandise...
-
Stabler Co.s projected March 31, 2011, balance sheet follows. Additional information about the company is as follows: ¢ Expected sales for April and May are $240,000 and $260,000, respectively....
-
Bernardino Santos-Rodiguez was piloting a boat owned by his friend, Raul Viera-Torres, in waters near Guayama, Puerto Rico. The boat was equipped with a hydraulic steering system manufactured by...
-
Exchange Corp. is a company that acts as a facilitator in tax-favored real estate swaps. Such swaps, know as 1031 exchanges, permit participants to avoid some or all of the capital gains taxes that...
-
(Parent reference for BST) Revise TreeNode by adding a reference to a nodes parent, as shown below: BinaryTree.TreeNode #element: T #left: TreeNode #right: TreeNode #*parent: TreeNode Create a test...
-
The electrodeposition of copper can be used to determine the copper content of a sample. The sample is dissolved to produce Cu 2+ (aq), which is electrolyzed. At the cathode, the reduction half-cell...
-
(A) K sp for AgCl = 1.8 x 10 -10 . What would be the measured E cell for the voltaic cell in Example 19-11 if the contents of the anode half-cell were saturated AgCl(aq) and AgCl(s)? Example 19-11...
-
Assume that the following data describe the condition of commercial banking system: Total reserve:..................$80 billion Transaction deposit.........$700 billion Cash held by public:...........
-
Confirm the yield to maturity . Are there cases where the the bondholder will not receive this yield? Give 2 examples. Bond Information Maturity Date: 11/15/2050 Callable? Yes Coupon: 3.50% Price...
-
Explain Austin and Seitanidi's concept of collaboration and its relevance to nonprofit performance and social impact. Is their framework useful for evaluating options for nonprofit-to-nonprofit...
-
The corporate treasurer of a US multinational receives a fax on 21 February from its European subsidiary. The sub-sidiary will transfer e 10 million to the parent company on 16th August. The...
-
How are producers able to make nutrients that consumers are able to eat?
-
Down Under Boomerang, Inc., is considering a new 3-year expansion project that requires an initial fixed asset investment of $2.18 million. The fixed asset will be depreciated straight-line to zero...
-
Do you agree that the efficient market hypothesis is dead? And if EMH is no longer valid, are the valuation models no longer an appropriate point of study?
-
How is use of the word consistent helpful in fraud reports?
-
Since the club opened, a major concern has been the pool facilities. Although the existing pool is adequate, Mindy, Oscar, and Lori all desire to make LifePath a cutting-edge facility. Until the end...
-
Mr. Marinos purchase of LifePath Fitness was done through his business. The investment has always been accounted for using the cost method on his firms books. However, early in 2012 he decided to...
-
The annual report of PepsiCo. is presented in Appendix A. Instructions (a) See Note 1 to the financial statements and indicate what the consolidated financial statements include. (b) Using PepsiCos...
-
6. Two runners are running the curve for the 200 m. The runner in lane 8 has a velocity of 8 m/s. the radius of her curve is 45 meters. The runner in lane 1 has a radius of 36 meters. If the two...
-
A teaching assistant builds a parallel-plate capacitor with adjustable spacing between the plates. When the plates are at their initial separation, the capacitance is 9.00 F. (a) At this capacitance,...
-
How much current flows through a 0.55-mm-diameter copper wire if the magnitude of the electric field in it is 0.015 V/m?

Study smarter with the SolutionInn App


