Refer to the discussion of the Leclanch cell. (a) Combine the several equations written for the operation
Question:
Refer to the discussion of the Leclanché cell.
(a) Combine the several equations written for the operation of the Leclanché cell into a single overall equation.
(b) Given that the voltage of the Leclanché cell is 1.55 V, estimate the electrode potentials, E, for each of the half cell reactions. Why are your values only estimates?

Figure 19.14
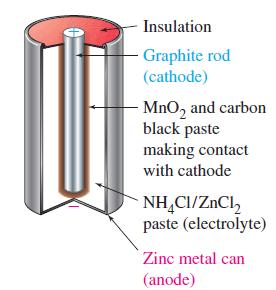
Transcribed Image Text:
The Leclanché (Dry) Cell The most common form of voltaic cell is the Leclanché cell, invented by the French chemist Georges Leclanché (1839-1882) in the 1860s. Popularly called a dry cell, because no free liquid is present, or flashlight battery, the Leclanché cell is diagrammed in Figure 19-14. In this cell, oxidation occurs at a zinc anode and reduction at an inert carbon (graphite) cathode. The electrolyte is a moist paste of MnO2, ZnCl2, NH4Cl, and carbon black (soot). The maximum cell voltage is 1.55 V. The anode (oxidation) half-cell reaction is simple. Oxidation: Zn(s) →→→ Zn²+ (aq) + 2 e The reduction is more complex. Essentially, it involves the reduction of MnO2 to compounds having Mn in a +3 oxidation state, for example, Reduction: 2 MnO₂ (s) + H₂O(1) + 2 e¯ Mn₂O3(s) + 2OH(aq) An acid-base reaction occurs between NH4+ (from NH4Cl) and OH¯. NH4+ (aq) + OH(aq) NH3(g) + H₂O(1) A buildup of NH3(g) around the cathode would disrupt the current because the NH3(g) adheres to the cathode. That buildup is prevented by a reaction between Zn²+ and NH3(g) to form the complex ion [Zn(NH3)2]²+, which crys- tallizes as the chloride salt. → Zn²+ (aq) + 2NH3(g) + 2Cl(aq) [Zn(NH3)2]Cl2(s) The Leclanché cell is a primary cell; it cannot be recharged. This cell is cheap to make, but it has some drawbacks. When current is drawn rapidly from the cell, products, such as NH3, build up on the electrodes, causing the voltage to drop. Also, because the electrolyte medium is acidic, zinc metal slowly dissolves. A superior form of the Leclanché cell is the alkaline cell, which uses NaOH or KOH in place of NH4Cl as the electrolyte. The reduction half-cell reaction is the same as that shown above, but the oxidation half-cell reaction involves the formation of Zn(OH)2(s), which can be thought of as occurring in two steps. Zn(s) Zn²+ (aq) + 2 e Zn²+ (aq) + 2OH(aq) Zn(OH)2(s) Zn(s) + 2OH(aq) → Zn(OH)2(s) + 2 e The advantages of the alkaline cell are that zinc does not dissolve as readily in a basic (alkaline) medium as in an acidic medium, and the cell does a better job of maintaining its voltage as current is drawn from it.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)

Answered By

Brown Arianne
Detail-oriented professional tutor with a solid 10 years of experience instilling confidence in high school and college students. Dedicated to empowering all students with constructive feedback and practical test-taking strategies. Effective educator and team player whether working in a school, university, or private provider setting. Active listener committed to helping students overcome academic challenges to reach personal goals.
4.60+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Refer to the discussion of the automobiles in the section on "TradingOff Conflicting Objectives: The Basics." We discussed switching first from the Standard to the Norushi, and then from the Norushi...
-
Refer to the discussion of beta values and market models in Problem 13.49 on page 553. The S&P 500 Index tracks the overall movement of the stock market by considering the stock prices of 500 large...
-
Refer to the discussion of Benfords law in Exercise 7.25. While this may seem like a curious oddity, researchers have developed some important applications for these proportions. One involves...
-
You are developing an industrial building with a gross building area of 150,000 sf. The building efficiency ratio is 75%. The market gross rent is $25 psf. The vacancy rate is 5%; the cap rate is 5%;...
-
Discuss the problems that might be encountered in having an expatriate sales manager supervising foreign salespeople.
-
The following data were taken from recent annual reports of Trump Hotels and Casino Resorts, Inc., which owns and operates casino-based entertainment resorts in Atlantic City, New Jersey. a....
-
Repeat the calculations of Example 6.3, but for \(80 \mathrm{~mol} \%\) of the liquid distilled. Data From Example 6.3:- Suppose the liquid of Example 6.1 [50 mol% n-heptane (A), 50 mol% n-octane...
-
Bradburn Corporation was formed 5 years ago through a public subscription of common stock. Daniel Brown, who owns 15% of the common stock, was one of the organizers of Bradburn and is its current...
-
L = {w| L2 = {w| Two languages L1 and L2 are defined by symbols = {0, 1}, written as w contains 00 as a sub-string and the length of w is even number} w begins with 0 and ends with 1} (i) Write two...
-
For the voltaic cell, (a) what isE cell initially? (b) As the cell operates, willE cell increase, decrease, or remain constant with time? Explain. (c) What will beE cell when [Ag + ] has increased to...
-
For the voltaic cell, (a) What is E cell initially? (b) If the cell is allowed to operate spontaneously, willE cell increase, decrease, or remain constant with time? Explain. (c) What will beE cell...
-
Sam Snead has formulated the following list of statements about contemporary developments in managerial accounting. 1. Just-in-time processing results in a push approach; that is, raw materials are...
-
After graduation from METU you will start working as an operations manager at an electronics manufacturer that faces assembly-line balancing problems. You have been told that the firm needs to...
-
An organization's portfolio investment process consists of the following activities performed once a year: ideas for projects are collected along with their projected benefits, development managers...
-
You just acquired a mortgage in the amount of $249,500 at 5.75 percent interest, compounded monthly. Equal payments are to be made at the end of each month for thirty years. How much of the first...
-
An atom has a diameter of 3.00 A and the nucleus of that atom has a diameter of 7.50 10 5 . Determine the fraction of the volume of the atom that is taken up by the nucleus. Assume the atom and the...
-
1. Why are people in India prosperous everywhere else besides India? What is the evidence? 2. Why are people from Hong Kong so prosperous as compared to everywhere else? What is the evidence? 3. What...
-
Following are the individual financial statements for Gibson and Davis for the year ending December 31, 2013: Gibson acquired 60 percent of Davis on April 1, 2013, for $528,000. On that date,...
-
A firm offers two products for sale. The marginal cost of one product is new zero once the first unit has been produced. The marginal cost of the other product rises as output rises. What would be...
-
Based on the data presented in Exercise 25-21, assume that Ohio Glass wanted to price all products so that they produced the same profit potential as the highest profit product. Thus, determine the...
-
Cardio Care Inc. manufactures stationary bicycles and rowing machines. The products are produced in the Fabrication and Assembly production departments. In addition to production activities, several...
-
Titan Industries manufactures two types of electrical power units, custom and standard, which involve four overhead activities?production setup, procurement, quality control, and materials...
-
Below is a two-period price tree of ABC Stock. A European call option on this stock has a strike price of $75 and the risk-free rate is 5% per sub-period. Given that the value of u is 1.22, calculate...
-
Delta Bank plans to issue 10 000, 10-year, 15% coupon bonds. Each bond will be issued at a par value of R1 000. To make the bonds attractive to investors, the bank plans to issue them at a discount...
-
How do you implement advanced cryptographic techniques, such as homomorphic encryption or zero-knowledge proofs, to ensure the verifiability and authenticity of data, thereby maintaining data...

Study smarter with the SolutionInn App


