Substances that dissolve in water generally do not dissolve in benzene. Some substances are moderately soluble in
Question:
Substances that dissolve in water generally do not dissolve in benzene. Some substances are moderately soluble in both solvents, however. One of the following is such a substance. Which do you think it is and why?
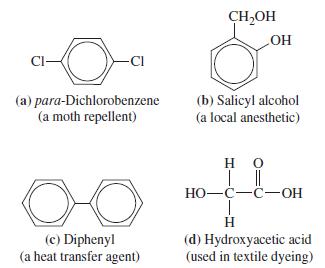
Transcribed Image Text:
CI- (a) para-Dichlorobenzene (a moth repellent) OO (c) Diphenyl (a heat transfer agent) CH₂OH LOH (b) Salicyl alcohol (a local anesthetic) HO | || HO-C-C-OH T H (d) Hydroxyacetic acid (used in textile dyeing)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The answer is b Salicyl alcohol Substances that dissolve in water generally do not dissolve in benze...View the full answer

Answered By

Tobias sifuna
I am an individual who possesses a unique set of skills and qualities that make me well-suited for content and academic writing. I have a strong writing ability, allowing me to communicate ideas and arguments in a clear, concise, and effective manner. My writing is backed by extensive research skills, enabling me to gather information from credible sources to support my arguments. I also have critical thinking skills, which allow me to analyze information, draw informed conclusions, and present my arguments in a logical and convincing manner. Additionally, I have an eye for detail and the ability to carefully proofread my work, ensuring that it is free of errors and that all sources are properly cited. Time management skills are another key strength that allow me to meet deadlines and prioritize tasks effectively. Communication skills, including the ability to collaborate with others, including editors, peer reviewers, and subject matter experts, are also important qualities that I have. I am also adaptable, capable of writing on a variety of topics and adjusting my writing style and tone to meet the needs of different audiences and projects. Lastly, I am driven by a passion for writing, which continually drives me to improve my skills and produce high-quality work.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Substances that dissolve in water generally do not dissolve in benzene. Some substances are moderately soluble in both solvents, however. One of the following is such a substance. Which do you think...
-
When a pure substance is placed in contact with water, there are three possible outcomes. The substance may do nothing that is, the substance does not dissolve and no visible change takes place. The...
-
a. Find b,,b,,c, and e, such that the maximization problem of the dual canonical tableau X, x, -1 10 b, 0 2 2 b. -1 3-f =5, = 5, =8 is in basic solution optimal form and such that both the...
-
Radon levels in a building interior are recorded at 8 pCi/L. What are the U.S. Environmental Protection Agency recommendations for this level?
-
Delby Industries has manufactured prefabricated houses for over 20 years. The houses are constructed in sections to be assembled on customers lots. Delby expanded into the precut housing market when...
-
The three forces act on the pipe assembly. If F1 = 50 N and F2 = 80 N, replace this force system by an equivalent resultant force and couple moment acting at O. Express the results in Cartesian...
-
The codon change (Gly-12 to Val-12) in human H-ras that converts it to oncogenic H-ras has been associated with many types of cancers. For this reason, researchers would like to develop drugs to...
-
Ueker Company is considering three capital expenditure projects. Relevant data for the projects are as follows. Annual income is constant over the life of the project. Each project is expected to...
-
6. Suppose the plates of a square parallel-plate capacitor are placed 2.0mm apart. If the area of each plate is 0.5cm, how many excess electrons will be on the negative plate if a voltage of 1.5V is...
-
Do you think that Henrys law works better for solutions of HCl(g) in benzene, C 6 H 6 , than it does for solutions of HCl(g) in water? If so, why?
-
(A) A 16.00% aqueous solution of glycerol, HOCH 2 CH(OH)CH 2 OH, by mass, has a density of 1.037 g/mL. What is the mole fraction of glycerol in this solution? (B) A 10.00% aqueous solution of...
-
Find the critical value(s) and rejection region(s) for the type of z-test with level of significance a. Include a graph with your answer. Two-tailed test, = 0.12
-
Examine the range of theories underpinning job design, noting any shortcomings.
-
Explain what is meant by: (a) attribution theory; and (b) impression management.
-
Highlight signifcant points in the debate about the impact of new technology on job design, in particular recent developments in AI and automation.
-
Why is reinforcement theory a learning theory considered in the context of motivation?
-
After consultation with its employees, Sunshine Company decides to switch the employee pay period to biweekly instead of weekly. Sunshines weekly payroll is $925,000. The annual cost of funds is 12%....
-
Following are Nintendos revenue and expense accounts for a recent calendar year (yen in millions). Prepare the companys closing entries for its revenues and its expenses. Net sales . . . . . . . . ....
-
When the Department of Homeland Security created a color-coded system to prepare government officials and the public against terrorist attacks, what did it do right and what did it do wrong?
-
Company A has issued a single zero-coupon bond maturing in 10 years. Company B has issued a coupon bond maturing in 10 years. Explain why it is more complicated to value Bs debt than As.
-
Company X has borrowed $150 maturing this year and $50 maturing in 10 years. Company Y has borrowed $200 maturing in five years. In both cases asset value is $140. Why might X not default while Y...
-
It has been suggested that the Fisher theory is a tautology. If the real rate of interest is defined as the difference between the nominal rate and the expected inflation rate, then the nominal rate...
-
K The Grand Inc. allocates FMOH to products on the basis of standard direct manufacturing labour-hours. For the year, FMOH was budgeted at $3.40 per direct manufacturing labour-hour (DMLH). Actual...
-
Ace Architects employs two architects, each having a different area of specialization. Caitlin specializes in industrial commercial construction and Zachary specializes in residential construction....
-
Reddington Enterprises is considering the two capital budeting projects with the following cash flows that have a WACC of 11%. Year Redd Wine Vineyards Kaplan Cleaners -200,000 -200,000 1 50,000...

Study smarter with the SolutionInn App


