The conversion of O 2 (g) to O 3 (g) can be accomplished in an electric discharge,
Question:
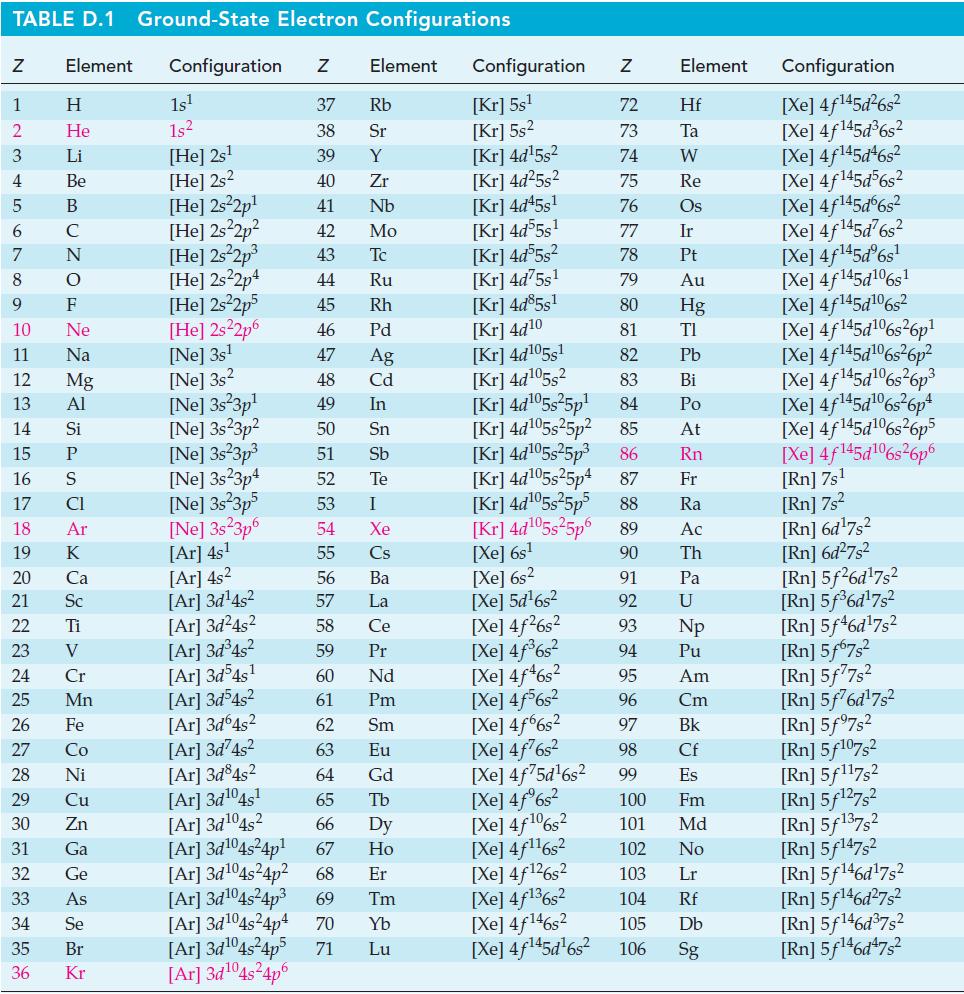
The conversion of O2(g) to O3(g) can be accomplished in an electric discharge, 3 O2(g) → 2 O3(g). Use a bond dissociation energy of 498 kJ mol–1 for O2(g) and data from Appendix D to calculate an average oxygen-to-oxygen bond energy in O3(g).
Transcribed Image Text:
TABLE D.1 Ground-State Electron Configurations Element Configuration Z Z 1 2 3 4 5 6 7 8 9 HIG&LUZONSUZ SE> 0 ≤ 2 3 2 3 5 3 3 4 8 5 2 He 10 11 12 13 14 15 16 17 18 19 20 21 Sc 22 23 24 25 Mn Mg 26 27 28 Ni 29 30 Zn 31 32 33 34 35 36 Ga Ge 1s¹ 1s² [He] 2s¹ [He] 2s2 [He] 2s²2p¹ [He] 2s²2p² [He] 2s²2p³ [He] 2s22p4 [He] 2s²2p5 [He] 2s²2p6 [Ne] 3s¹ [Ne] 3s2 [Ne] 3s 3p¹ [Ne] 3s 3p² [Ne] 3s²3p³ [Ne] 3s23p4 [Ne] 3s²3p5 [Ne] 3s23p6 [Ar] 4s¹ [Ar] 4s² [Ar] 3d¹4s² [Ar]3d²4s² [Ar]3d³4s² [Ar] 3d54s¹ [Ar] 3d³4s² [Ar]3d64s² [Ar] 3d²4s² [Ar]3d845² [Ar] 3d¹04s¹ [Ar] 3d¹04s2 [Ar] 3d¹04s²4p¹ Element 37 Rb 38 Sr 39 Y 40 Zr 41 Nb 42 Mo 43 Tc 44 Ru 45 Rh 46 Pd 47 Ag 48 Cd 49 In 50 Sn 51 Sb 52 Te 53 54 55 56 57 58 59 60 61 62 63 64 I Xe Cs Ba La Ce Pr Nd Pm Sm Eu Gd 65 Tb 66 67 Dy Ho Er [Ar] 3d¹04s²4p² 68 [Ar] 3d¹04s²4p³ 69 Tm [Ar] 3d¹04s²4p4 70 Yb [Ar]3d¹04s²4p5 71 Lu [Ar] 3d¹04s²4p6 Configuration Z [Kr] 5s¹ [Kr] 5s² [Kr] 4d¹5s² [Kr] 4d²5s² [Kr] 4d45s¹ [Kr] 4d55s¹ [kr] 4d55s² [Kr] 4d75s¹ [Kr] 4d85s1 [Kr] 4d10 [Kr] 4d105s1 [kr] 4d¹05s² [kr] 4d¹05s²5p¹ [Kr] 4d¹05s25p² [kr] 4d¹05s²5p³ [Kr] 4d¹05s25p4 [Xe] 6s² [Xe] 5d¹6s² [Xe] 4f²6s² [Xe] 4f³6s² [Xe] 4f46s2 [Xe] 4f6s2 [Xe] 4f6s2 [Xe] 4f²6s² [Xe] 4f75d¹6s² [Xe] 4f%s2 [Xe] 4f106s2 [Xe] 4f¹¹6s² NRNKERKR [Xe] 4f126s2 [Xe] 4f136s2 [Xe] 4f146s2 [Xe] 4f¹45d¹6s² 72 Hf 73 Ta W 74 75 Re 76 Os 77 Element 78 79 80 81 82 83 84 85 86 87 Fr [kr] 4d¹05s²5p5 88 Ra [Kr] 4d¹05s²5p6 89 Ac [Xe] 6s¹ 90 Th 91 92 93 94 95 96 97 98 99 Ir Pt Au Hg TI Pb Bi Po At Rn Pa U Np Pu Am Cm Bk Cf Es 100 Fm 101 Md 102 No 103 Lr 104 Rf 105 Db 106 Sg Configuration [Xe] 4f¹45d²6s² [Xe] 4f145d³6s² [Xe] 4f145d46s2 [Xe] 4f145d56s2 [Xe] 4f145d6s2 [Xe] 4f¹45d²6s² [Xe] 4f¹45dº6s¹ [Xe] 4f145d106s1 [Xe] 4f145d106s2 [Xe] 4f145d6s26p* [Xe] 4f145d106s36p? [Xe] 4f145d16s?6p3 [Xe] 4f145d6s®6p* [Xe] 4f145d16s26p5 [Xe] 4f145d106s26p6 [Rn] 7s¹ [Rn] 7s² [Rn] 6d¹7s² [Rn] 6d²7s² [Rn] 5f26d¹7s² [Rn] 5f³6d¹7s² [Rn] 5f46d¹7s2 [Rn] 5f67s² [Rn] 5f77s² [Rn] 5f76d¹7s² [Rn] 5f97s2 [Rn] 5f107,2 [Rn] 5f117s2 [Rn] 5f¹27s² [Rn] 5f137,2 [Rn] 5f147s2 Rn] 5f¹46d¹7s² [Rn] 5f¹46d²7s² [Rn] 5f¹46d³7s2 [Rn] 5f¹46d47s²
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
3021 20 Bond dissociation energies AH dissociation AH ...View the full answer

Answered By

PULOK NATH
I HAVE A EXPERIENSCED AROUND 15 YEARS ON COMPUTER APPLICATION ESPECIALLY OFFICE AUTOMATION ETC. STILL I AM TEACHING ABACUS INDIA PVT. LTD AS A FACULTY OF BASIC MATHEMATICS FROM 2014 ONWARDS.
0.00
0 Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Estimate the average bond energy in O 3 (g) from the structure and data in Table 10.3. Compare this result with that of Exercise 35. Table 10.3 Exercise 35 The conversion of O 2 (g) to O 3 (g) can be...
-
Resonance energy is the difference in energy between a real moleculea resonance hybridand its most important contributing structure. To determine the resonance energy for benzene, we can determine an...
-
Use the following data and data from Appendix D to determine the quantity of heat needed to convert 15.0 g of solid mercury at -50.0 C to mercury vapor at 25 C. Specific heats: Hg(s), 24.3 J mol -1 K...
-
In order to evaluate lim f(a+h)-f(), it is necessary to evaluate f(a + h). h xa For f(x) = x 3, f(a+h) =
-
Shiraz Company invested $12,100 in a mutual fund on August 1. By December 31, the value of the mutual fund had declined to $8,800, and the company did not sell any portion of the mutual fund during...
-
What is the nature and purpose of a letter of representations? Comment on the quality or strength of the audit evidence yielded by a letter of representations.
-
Based on the design, briefly discuss the data collection procedures to be used. Be sure to include the area rea of focus and targeted sample as part of these procedures. Develop a hypothetical...
-
As audit partner for Grupo and Rijo, you are in charge of reviewing the classification of unusual items that have occurred during the current year. The following material items have come to your...
-
The given code starts by initializing some sets and functions f , g , and h . Block 1 is a function ( called hasInverse ) that takes as input another function ( called func ) , and two sets ( D and T...
-
The natural abundance of O 3 in unpolluted air at ground level is about 0.04 parts per million (ppm) by volume. What is the approximate partial pressure of O 3 under these conditions, expressed in...
-
Without performing detailed calculations, determine which of the following compounds has the greatest percent oxygen by mass: dinitrogen tetroxide, aluminum oxide, tetraphosphorus hexoxide, or carbon...
-
In Problems 2328, define the variable and translate the sentence into an inequality. The annual salary is at least $65,000.
-
When it comes to personal selling, the term win-win gets thrown around so much that it has become a clich. But at Procter & Gamble, the selling concept that the company benefits only if the customer...
-
Social media networks seem numberless these days, but only an elite few that started in the early days of the World Wide Web still survive. One of those is LinkedIn, a social media site that debuted...
-
When people think of buying and selling things online locally, most think automatically of Craigslist, the classified ad marketplace that has dominated that business for the past two decades. But as...
-
China has emerged as an enormous social media market. With more than 1.35 billion people and 635 million internet users, internet usage in China is growing explosively at about 30 percent annually....
-
Facebook has more than 2 billion monthly active users. What started as an online social network allowing people to connect with each other has transformed into a behemoth media mogul and a real...
-
Evander, an officer in the Marine Corps, was appointed commanding officer of the Marine training base in Beaufort, S.C. As is customary in connection with changes in command, he hosts a party at his...
-
Which should drive action planning more, strengths or weaknesses? That is, is it more important to build on your strengths or to reduce your weaknesses? Explain.
-
Ndon Company has the following internal control procedures over cash disbursements. Identify the internal control principle that is applicable to each procedure. (a) Company checks are prenumbered....
-
Jay Bauer is uncertain about the control features of a bank account. Explain the control benefits of (a) A checking account and (b) A bank statement.
-
The following reconciling items are applicable to the bank reconciliation for Gratz Co. Indicate how each item should be shown on a bank reconciliation (a) Outstanding checks. (b) Bank debit...
-
Following an event, the price of maple syrup for pancakes decreases. This will result in: An increase in the quantity demanded of maple syrup and an increase in the quantity demanded of pancakes. An...
-
1. Identify the music excerpt you selected. (.5) C agliostro in Wien - Johann Strauss II 2. Identify the film selected for this assignment. (.5) San Andreas (2015) - Tsunami scene - Pure Action 3....
-
As a response to inflation, in March 2022, the Federal Reserve approved its first interest rate hike since December 2018. However, inflation still remained very high and piked in June 2022. The last...

Study smarter with the SolutionInn App


