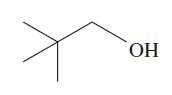
The molecule in the next column is 2,2-dimethylpropan-1-ol, a primary alcohol. Because no H atoms are bonded
Question:
The molecule in the next column is 2,2-dimethylpropan-1-ol, a primary alcohol. Because no H atoms are bonded to the β carbon atom in this molecule, dehydration seems unlikely. However, when 2,2-dimethylpropan-1-ol is heated with an acid catalyst, 2-methylbut-2-ene is obtained. Suggest a mechanism that shows how 2-methylbut-2-ene is formed.
Transcribed Image Text:
HO
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
The mechanism for the formation of 2methylbut2ene from 22dimethylpropan1ol involves an acidcataly...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Hypophosphorous acid, H3PO2, has the structure (HO)PH2O, in which two H atoms are bonded to the P atom, and one H atom is bonded to an O atom. For each bond to an H atom, decide whether it is polar...
-
Phosphorous acid, H3PO3, has the structure (HO)2PHO, in which one H atom is bonded to the P atom, and two H atoms are bonded to O atoms. For each bond to an H atom, decide whether it is polar or...
-
Give two examples of saturated hydrocarbons. How many other atoms are bonded to each carbon in a saturated hydrocarbon?
-
Identify each of the equations as representing either a circle, a parabola, an ellipse, a hyperbola, or none of these. (x + 1) 2 + (y + 1) 2 = 2(x + y + 1)
-
Jones Custom Furniture Manufacturing, Inc., made the following estimates at the beginning of the year, 2012: Budgeted direct labor costs . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
A1 Vineyard Fruit Company sells premium-quality oranges and other citrus fruits by mail order. Protecting the fruit during shipping is important so the company has designed and produces shipping...
-
Refer to the information in Exercise 22-12. Assume that each of the companys divisions has a required rate of return of 7%. Compute residual income for each division. Data From Exercise 22-12 A food...
-
Condensed financial data of Minnie Hooper Company are shown below. Additional information: 1. New plant assets costing $149,000 were purchased for cash during the year. 2. Investments were sold at...
-
Consider the acceleration of a good sports car in real life . (1) Pick a certain car model, which could be the one you are driving, or the one you wish you were --- and search online for its...
-
Draw the structures of the products of each of the following reactions: (a) Propene + hydrogen (Pt, heat); (b) Butan-2-ol + heat (in the presence of sulfuric acid).
-
Give the structures of the main organic product(s) in each of the following reactions. (a) (b) (c) + Br CC14
-
Why is the auditor concerned about the impact of possible impairment of value on the fixed assets of audit clients? Summarize the accounting guidance regarding impairment. What are the related...
-
(b) Complete a table of DH parameters for the robot shown below. Explain your reasoning. In your answer provide a description of each of the parameters. 0 0.3m 0.35m X3 23 0.25m [7 marks] 0.1m 0.2m
-
1. (a) Calculate P. (Assume suitable values for the required parameters). Atmosphere AIR P=? h=15 mm (2 M) (b) An ideal gas with Cv=5/2 R, at 700 K and 1.5 bar pressure receives 25 kJ of heat during...
-
2.37 The output of an instrument is often a waveform. With the goal of developing a numerical measure of closeness, scientists asked 11 experts to look at two waveforms on the same graph and give a...
-
1. a) Rank the magnitudes of the diffusion coefficients from greatest to least for the following systems: N in Fe at 600 C Cr in Fe at 600 C N in Fe at 1000 C Cr in Fe at 1000 C Now justify this...
-
An engineering component is made of the Boron carbide (B4C) ceramic of Table 3.10. The most severely stressed point is subjected to the following state of stress: 0x = 35, oy = 21, Txy = 7, and o =...
-
Julie Krenzke worked as a financial services manager for Lindsay Lexus from March 2002 until October 2004. Krenzke was very good at her job when she was present, but she was often absent due to...
-
APC16550D UART has a clock running at18.432 MHz and its baud rate is set to 2000.Determine the HEX contents of its DLM and DLL registers. Please can you explain step by step and in detail how you get...
-
Suppose a 10-year, 10 percent, semiannual coupon bond with a par value of $1,000 is currently selling for $1,135.90, producing a nominal yield to maturity of 8 percent. However, the bond can be...
-
If you bought this bond, do you think you would be more likely to earn the YTM or the YTC? Why?
-
Write a general expression for the yield on any debt security (rd) and define these terms: real risk-free rate of interest (r*), inflation premium (IP), default risk premium (DRP), liquidity premium...
-
1. Given y == -tan 0, 03 a. Sketch the graph for the given interval, show & label any asymptotes. (2 marks) b. Draw the line y = -1 on the graph in part a, and state the coordinates for the points...
-
Flynn Incorporated acquires 100 percent of the outstanding voting shares of Macek Company on January 1, 2024. To obtain these shares, Flynn pays $400 cash (in thousands) and issues 10,000 shares of...
-
Given a collection of unencrypted plain-text files, and the MD5 hash of one of the files, build a solution to identify which of the plain-text files matches the hash given to you. What kind of code...

Study smarter with the SolutionInn App


