The situation pictured in Figure 6-10(a) is changed to that in Figure 6-10(b). What is the gas
Question:
The situation pictured in Figure 6-10(a) is changed to that in Figure 6-10(b). What is the gas pressure in Figure 6-10(b)?
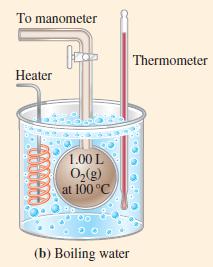
Figure 6-10(a)
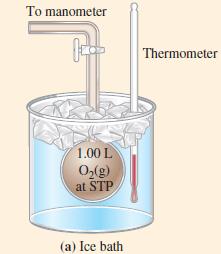
Figure 6-10(b)
Transcribed Image Text:
To manometer 1.00 L O₂(g) at STP (a) Ice bath Thermometer
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Analyze Identify the quantities in the general gas equation that remai...View the full answer

Answered By

Mary Njunu
I posses Vast, diversified knowledge and excellent grammar as a result of working in ACADEMIC WRITING for more than 5 years. I deliver work in various disciplines with assurance of quality work. I purpose at meeting the clients’ expectations precisely. Let’s work together for the best and phenomenal grades.
4.90+
928+ Reviews
2551+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
1. How strong are the competitive forces confronting J. Crew in the market for specialty retail? Do a [Michael Porter] five-forces analysis to support your answer. (see chapter 3 in the textfor...
-
In the situation pictured in question 3, if there is a frictional force opposing the motion of the block, does this frictional force do work on the block? Explain.
-
In the situation pictured in question 28, is the magnitude of the velocity important to the success of the shot? Explain. Data from problem 28 A Q28 Diagram
-
A jet is traveling westward with the sun directly overhead (the jet is on a line between the sun and the center of the Earth). How fast must the jet fly in order to keep the sun directly overhead?...
-
What are the four perspectives of the balanced scorecard?
-
On January 1, 2015, Perriman Company sold equipment for cash and leased it back. As seller-lessee, Perriman retained the right to substantially all of the remaining use of the equipment. The term of...
-
The wreck of the S.S. Central America waited 130 years at the bottom of the ocean for someone to come along and claim its trove of gold. Thomas G. Thompson, funded by a multitude of investors, was...
-
Performance reports and evaluation Vance Corporation has four divisions: the assembly division, the processing division, the machining division, and the packing division. All four divisions are under...
-
Corporate taxation in South Africa is too high and has negative implications for economic growth and our competitiveness with our main trading partners." Do you agree with this statement? Why (not)?...
-
Without performing an actual calculation, indicate which has the greater u rms , He(g) at 1000 K or H 2 (g) at 250 K.
-
(A) A 1.00 mL sample of N 2 (g) at 36.2 C and 2.14 atm is heated to 37.8 C, and the pressure changed to 1.02 atm. What volume does the gas occupy at this final temperature and pressure? (B) Suppose...
-
Suppose that f(x) = x 2 + 4x - 21 (a) What is the vertex of f? (b) What are the x-intercepts of the graph of f? (c) Solve f(x) = -21 for x. What points are on the graph of f? (d) Use the information...
-
Briefly Explain and Indicate the type of ruling or decision that has been made by the Commissioner and the extent to which it is binding on the taxpayers involved.? The following tax rulings were...
-
How much is the deductible pension contributions for 2021? How much is Jiho Corporation's income tax payable? Consider the following information regarding a corporation's pension contributions: ...
-
What recent news should I refer to when I try to teach inflation such as measurement, cause (demand-pull inflation and cost-push inflation), expected inflation? Could you give me some suggestions?
-
Explain the different stakeholders within the financial markets and their priorities, highlighting where they are incompatible? How do the goals and methods which the FCA employ help to make...
-
1. A real property was transferred to a revocable trust with Carding as the beneficiary. The property earned a total of 900,000 gross rentals during the taxable period. The expenses attributable to...
-
The U. S. government spends over $33 billion on its Food Stamp program to pro-vide millions of Americans with the means to purchase food. These stamps are redeemable for food at over 160,000 store...
-
On March 31, 2018, Gardner Corporation received authorization to issue $30,000 of 9 percent, 30-year bonds payable. The bonds pay interest on March 31 and September 30. The entire issue was dated...
-
(Two Temporary Differences, Multiple Rates, Future Taxable Income) Flynn Inc. has two temporary differences at the end of 2010. The first difference stems from installment sales, and the second one...
-
(Two Differences, One Rate, First Year) The differences between the book basis and tax basis of the assets and liabilities of Morgan Corporation at the end of 2010 are presented below. It is...
-
(NOL Carry back and Carry forward, Valuation Account versus No Valuation Account) Sondgeroth Inc. reports the following pretax income (loss) for both financial reporting purposes and tax purposes....
-
A water filtration plant desired to filter 6000 gallons of water per hour through a sand filter bed to remove suspended matters and solids from water. Based on the data given, determine the...
-
A distillation column is used to separate benzene and toluene by distillation. The relative volatility for this system is approximately 2.5 with benzene the more volatile component. a) To find the...
-
4. Write the chemical equilibrium equations for the following three reactions, respectively. Indicate how the equilibrium constants are related: 2 NH3 + 3NO 3 HO + N 4 NH3 + 6NO 6 H2O + 5 N2 3 H2O +...

Study smarter with the SolutionInn App


