(A) A 1.00 mL sample of N 2 (g) at 36.2 C and 2.14 atm is heated...
Question:
(A) A 1.00 mL sample of N2(g) at 36.2 °C and 2.14 atm is heated to 37.8 °C, and the pressure changed to 1.02 atm. What volume does the gas occupy at this final temperature and pressure?
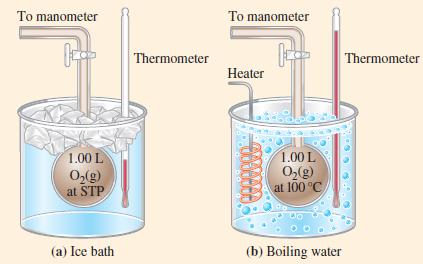
(B) Suppose that in Figure 6-10 we want the pressure to remain at 1.00 bar when the O2(g) is heated to 100 °C. What mass of O2(g) must we release from the flask?
Figure 6-10
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:





