Use 1 x 10 -13 cm as the approximate diameter of the spherical nucleus of the hydrogen-1
Question:
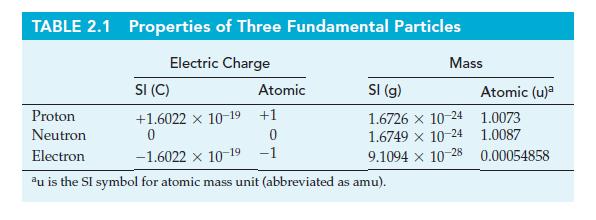
Use 1 x 10-13 cm as the approximate diameter of the spherical nucleus of the hydrogen-1 atom, together with data from Table 2.1, to estimate the density of matter in a proton.
Table 2.1
Transcribed Image Text:
TABLE 2.1 Properties of Three Fundamental Particles Electric Charge Atomic Mass SI (C) Proton +1.6022 x 10-19 +1 Neutron 0 0 Electron -1.6022 × 10-19 -1 au is the SI symbol for atomic mass unit (abbreviated as amu). SI (g) 1.6726 x 10-24 1.6749 × 10-24 9.1094 x 10-28 Atomic (u)² 1.0073 1.0087 0.00054858
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
Density d massg volume gcm mass g proton 16726x1...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
The hydrogen atom is composed of one proton in the nucleus and one electron, which moves about the nucleus. In the quantum theory of atomic structure, it is assumed that the electron does not move in...
-
The large magnetic fields used in MRI can produce forces on electric currents within the human body. This effect has been proposed as a possible method for imaging biocurrents flowing in the body,...
-
An atom has a very small nucleus surrounded by an electron cloud. Figure 2.1 represents the nucleus with a diameter of about 2 mm and describes the electron cloud as extending over 200 m. If the...
-
Each of the following passages may be plausibly criticized by some who conclude that it contains a fallacy, but each may be defended by some who deny that the argument is fallacious. Discuss the...
-
a. A vacant lot acquired for $75,000 is sold for $145,000 in cash. What is the effect of the sale on the total amount of the sellers? (1) Assets (2) Liabilities (3) Owner's equity? b. Assume that the...
-
Go to the library and find any data that you can of The Art of War by Sun Tzu (many other authors have interpreted this work so look for other authors of this same title). Relate the concept of...
-
During a storm, a snow drift is formed behind a snow fence as shown in Fig. P7.56. Assume that the height of the drift, \(h\), is a function of the number of inches of snow deposited by the storm,...
-
At September 30, 2014, the accounts of Park Terrace Medical Center (PTMC) include the following: Accounts Receivable ............... $ 141,000 Allowance for Bad Debts (credit balance) .....3,400...
-
5. Design a direct-mapped cache structure. The cache has the capacity of 8kB (kilo-byte), assuming 1 block is 512 bytes, and 1 valid bit, 1 tag bit. Calculate (S, E, B, m), draw and explain the cache...
-
Fluorine has a single atomic species, 19 F. Determine the atomic mass of 19 F. by summing the masses of its protons, neutrons, and electrons, and compare your results with the value listed on the...
-
William Prout (1815) proposed that all other atoms are built up of hydrogen atoms, suggesting that all elements should have integral atomic masses based on an atomic mass of one for hydrogen. This...
-
The publisher of a magazine gives his staff the following information: Current price ........................ $2.00 per issue Current sales ........... 150,000 copies per month Current total costs...
-
Mr. Arben is a very famous comedian in Europe. He is hired to fly all the way to Canton, Michigan by Professor's Watts to perform at her birthday party. He calls a week before and says, "I am...
-
Suppose that the U.S. Federal Reserve wishes to influence the value of the dollar to decrease inflation. If the Fed uses direct intervention to achieve this goal, it will seek to strengthen/weaken...
-
Confronting Intercultural Difficulties. How might you deal with any one or two of the following obstacles to intercultural communication? If you have the opportunity, share your responses with others...
-
A 115 kg hockey player, Adam, is skating east when he tackles a stationary 133 kg player, Bob. Afterward, they move at 1.35 m/s east. What was Adam's velocity before the collision.
-
1. Macie places Q1= 90C at the origin, Q2 = 50C at 40 cm from origin in positive x-direction and Q3 = -80C at 80 cm from origin in positive x direction. Find the force on Q2 due to the other two...
-
In order to estimate the mean 30-year fixed mortgage rate for a home loan in the United States, a random sample of 28 recent loans is taken. The average calculated from this sample is 5.25%. It can...
-
Why is it important to understand the macro-environment when making decisions about an international retail venture?
-
What are two potential tests that can be conducted to verify the CAPM? What are the results of such tests? What is rolls critique of CAPM tests?
-
Briefly explain the difference between the CAPM and the arbitrage pricing theory (APT).
-
Suppose you are given the following information. The beta of company, bi, is 0.9, the risk free rate, rRF, is 6.8%, and the expected market premium, rM-rRF, is 6.3%. Because your company is larger...
-
Although Hank is retired, he is an excellent handyman and often works part time on small projects for neighbors and friends. Last week his neighbor, Mike, offered to pay Hank $680 for minor repairs...
-
Manufacturers Southern leased high - tech electronic equipment from International Machines on January 1 , 2 0 2 4 . International Machines manufactured the equipment at a cost of $ 9 5 , 0 0 0 ....
-
For the past year, a firm had sales of $41,987, interest expense of $3,232, cost of goods sold of $16,500, selling and administrative expense of $11,000, and depreciation of $6,200. If the tax rate...

Study smarter with the SolutionInn App


