Question: Use data from Appendix D to calculate r H for the following reaction at 25 C. Fe2O3(s) + 3 CO(g) 2 Fe(s) + 3
Use data from Appendix D to calculate ΔrH° for the following reaction at 25 °C.
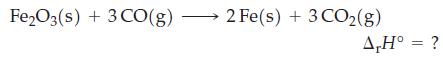
Fe2O3(s) + 3 CO(g) 2 Fe(s) + 3 CO(g) A,H = ?
Step by Step Solution
3.38 Rating (160 Votes )
There are 3 Steps involved in it
To calculate rH for the reaction at 25 C we need to use the enthalpy of f... View full answer

Get step-by-step solutions from verified subject matter experts


