Use data from Figure 20-12 to determine the temperature at which t 1/2 for the first-order decomposition
Question:
Use data from Figure 20-12 to determine the temperature at which t1/2 for the first-order decomposition of N2O5 in CCl4 is 2.00 h.
Figure 20-12
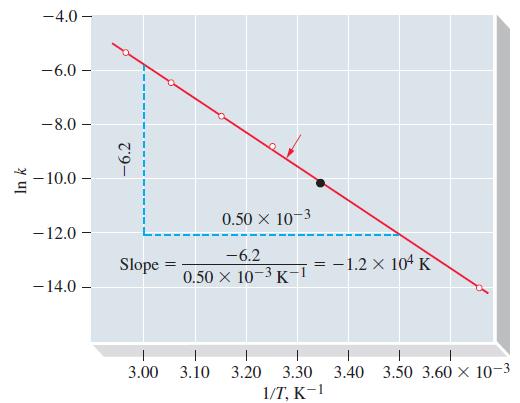
Transcribed Image Text:
In k -4.0 - -6.0- -8.0 -10.0- -12.0 - - 14.0 -6.2 Slope = 3.00 0.50 × 10-3 -6.2 0.50 x 10-3K-1 3.10 3.20 3.30 1/T, K-1 -1.2 x 104 K 3.40 3.50 3.60 × 10-3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
Analyze First find the rate constant k corresponding to a 200 h halflife This can be done by using t...View the full answer

Answered By

Morgan Njeri
Very Versatile especially in expressing Ideas in writings.
Passionate on my technical knowledge delivery.
Able to multitask and able to perform under pressure by handling multiple challenges that require time sensitive solution.
Writting articles and video editing.
Revise written materials to meet personal standards and satisfy clients demand.
Help Online Students with their course work.
4.90+
12+ Reviews
38+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
(A) What is the half-life of the first-order decomposition of N 2 O 5 at 75.0 C? Use data from Example 20-9. (B) At what temperature will it take 1.50 h for two-thirds of a sample of N 2 O 5 in CCl 4...
-
In Example 13-12, we used the vant Hoff equation to determine the temperature at which for the reaction 2 SO 2 (g) + O 2 (g) 2 SO 3 (g). Obtain another estimate of this temperature with data from...
-
The 50-mm-diameter cylinder is made from Am 1004-T61 magnesium and is placed in the clamp when the temperature is T 1 = 15°C. If the two 304-stainless-steel carriage bolts of the clamp each have...
-
Ha: μ1 > μ2 The null hypothesis is H0: μ1 = μ2 and the alternative hypothesis is as specified. We have provided data from a simple random...
-
Pockey Company has assembled the following market data, based on commercial real estate sales in the area in the past six months, which can be used to indirectly estimate the selling price of a...
-
The Home Finders Realty Corporation specializes in locating homes for prospective buyers. Home information is stored in a database and is to be shown on an inquiry display screen. Design a GUI...
-
According to the Supreme Court decision in Quill Corp. v. North Dakota, a state may not require a retailer with no physical presence in the state to collect and remit sales tax on sales made in the...
-
Main Street Ice Cream Company uses a plantwide allocation method to allocate overhead based on direct labor-hours at a rate of $3 per labor-hour. Strawberry and vanilla flavors are produced in...
-
1. Which of the following formulas can be considered an algorithm for computing the area of a triangle whose side lengths are given positive numbers a, b, and c? Explain these. a. Sp(p-a)(p - b)(pc),...
-
What is the approximate concentration of A remaining after 110 s in the (a) Zero-order, (b) First-order, (c) Second-order reaction? I Time, S 0 25 50 75 100 150 200 250 [A], M 1.00 0.78 0.61 0.47...
-
Use data from Table 20.3 to establish the order of reaction (20.7) with respect to HgCl 2 and C 2 O 4 2- and also the overall order of the reaction. Table 20.3 Reaction (20.7) TABLE 20.3 Kinetic Data...
-
(a) Define cost in relation to inventory. (b) What is meant by NRV? (c) Explain the rule of lower of cost and NRV for inventory.
-
Why are Religious organizations are not discussed in not-for-profit accounting?
-
What is the difference between traditional and activity-based product margins are primarily caused by a company's unit-level activities.?
-
Explain business entity convention, going concern convention and dual aspect convention and evaluate how important are they in the preparation of the three major financial statements?
-
What document provides prospective and general information about how PHI might be used or disclosed in the future?
-
Suppose a Greek tourist visits Abu Dhabi, and in particular a cloths market in Abu Dhabi Mall. The tourist bought one blue jean brand Wrangler for 159 AED and noticed that this particular jean is...
-
Distinguish between the gross and net methods of accounting for cash discounts.
-
Use the formula to determine the value of the indicated variable for the values given. Use a calculator when one is needed. When necessary, use the key on your calculator and round answers to the...
-
The trial balance for Pioneer Advertising Agency is shown in Illustration 3-3, p. 100. In lieu of the adjusting entries shown in the text at October 31, assume the following adjustment data. 1....
-
The income statement of Benning Co. for the month of July shows net income of $1,400 based on Service Revenue $5,500, Wages Expense $2,300, Supplies Expense $1,200, and Utilities Expense $600. In...
-
A partial adjusted trial balance of Sila Company at January 31, 2010, shows the following. InstructionsAnswer the following questions, assuming the year begins January 1.(a) If the amount in Supplies...
-
There are two concepts under confidentiality: Attorney-Client Privilege and the Rule of Confidentiality. Identify and describe at least four similarities and four differences.
-
Create and evaluate a scenario where, according to the Maryland Attorneys Rules of Professional Conduct, a conflict of interest might exist. The scenario must be detailed with names, actions, and a...
-
You know the prices of the following bonds, each of which pays coupons annually, and each of which will pay its next coupon in exactly one year: Name Coupon Maturity Price (years) A 1% 4 92.57 B 5% 4...

Study smarter with the SolutionInn App


