Use data from Figure 9-11 to predict the type of cubic unit cell adopted by (a) CaO;
Question:
Use data from Figure 9-11 to predict the type of cubic unit cell adopted by
(a) CaO;
(b) CuCl;
(c) LiO2 (the radius of the O2- ion is 128 pm).
Figure 9-11
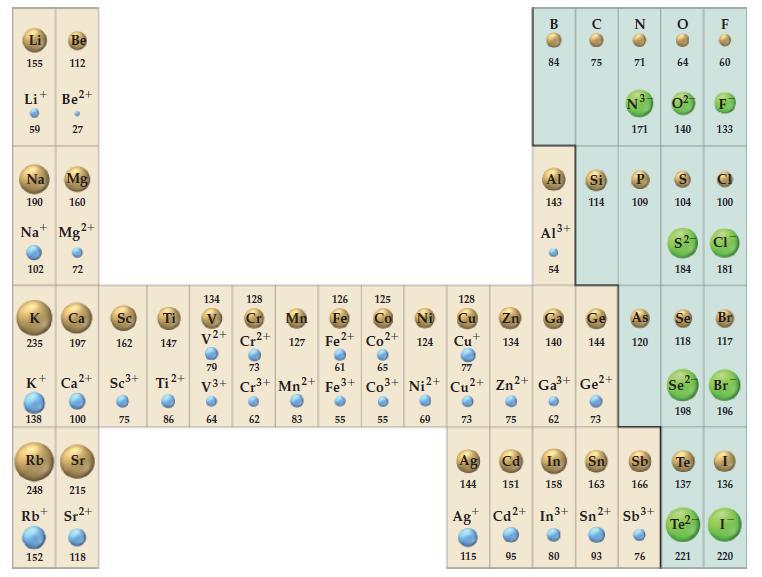
Transcribed Image Text:
155 59 Li+ Be²+ 102 K Na Mg 190 160 Na Mg2+ 235 Be 112 138 27 152 72 K+ Ca²+ Sc³+ Ca Sc Ti 197 162 147 100 Rb 248 215 Rb Sr²+ Sr 118 75 Ti ²+ 86 134 128 V2+ 79 V3+ 64 ២ Cr2+ 73 Cr³+ 62 126 Mn Fe 127 Fe2+ 2+ Mn2 83 61 Fe 3+ 55 125 Co2+ 65 Co3+ 55 Ni 124 Ni²+ 69 73 Ag 144 128 Cu Zn Ga Cu 134 140 с 115 75 B Cd 151 84 Al 143 A1³+ 95 54 77 Cu²+ Zn²+ Ga³+ Ge²+ 3+ 62 CO с 80 75 Ge 144 ● Si P 114 109 73 N ZO 93 71 N3 0² F 171 140 133 As 120 64 76 104 $2 In Sn Sb Te 158 163 166 137 + Ag Cd2+ In3+ Sn²+ Sb³+ 184 Se 118 Se 198 Te²- 60 221 100 CI 181 Br 117 Br 196 136 220
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
To predict the type of cubic unit cell adopted by a compound we need to compare the radii ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Use data from Figure 9-9 to predict the type of cubic unit cell adopted by (a) BaO; (b) CuI; (c) LiS 2 . (The radii of Ba 2+ and S 2 - ions are 135 and 198 pm, respectively.) Figure 9-9 Tne/ao 9 8 7...
-
The table below lists the ionic radii for the cations and anions in three different ionic compounds. Each compound has either the NaCl, CsCl, or ZnS type cubic structure. Predict the type of...
-
Mn crystallizes in the same type of cubic unit cell as Cu. Assuming that the radius of Mn is 5.6% larger than the radius of Cu and the density of Cu is 8.96 g/cm3, calculate the density of Mn.
-
Prerequisite: You will be using "utils.py" from Python 2 Assignment Task 1) In "utils.py," add a Python function called "calculate_fourier_coefficients" that calculates the coefficients of a Fourier...
-
Bishop Logging Company is a large, family-owned logging contractor formed in the low country of South Carolina. Bishop Logging has traditionally harvested pine timber. However, Bishop Logging began...
-
The load on a bolt consists of an axial pull of 10 KN together with a transverse shear force of s KN Find the diameter of bolt required according to 1. Maximum principal stress theory 2. Maximum...
-
Refer to Googles financial statements in Appendix A to compute its equity ratio as of December 31, 2015, and December 31, 2014. Data From Google Financial Statement Appendix A Google Inc....
-
For the past several years, Abby Brown has operated a part-time consulting business from her home. As of June 1, 2012, Abby decided to move to rented quarters and to operate the business, which was...
-
In general, should a country's policymakers be concerned if the country has trade imbalances with other countries? If your answer ispolicymakers "no", are there situations in which policy makers...
-
Potassium chloride has the same crystal structure as NaCl. Careful measurement of the internuclear distance between K + and Cl - ions gave a value of 314.54 pm. The density of KCl is 1.9893 g/cm 3 ....
-
Two views, a top and side view, for the unit cell for rutile (TiO 2 ) are shown here. (a) How many titanium atoms (blue) are in this unit cell? (b) How many oxygen atoms (red) are in this unit cell?
-
Why does tax planning analysis focus on the marginal tax rate?
-
Describe an example you have experience with of the principal-agent problem (for example, you have a desire for your children to listen in school and do well, while for them, goofing off at school...
-
Why do skilled professionals working in the same business often have significantly different forecasts and views of the future? Share an example from your own work experience at CVS Health where your...
-
Taylor Rules and Short-Run Exchange Rate movements for inflation targeting central banks. Consider the following Taylor Rule for the nominal short-term U.S. policy interest rate ($): = ( ) ()( )...
-
what are examples of how purchasing has influenced business within the last 10 years?
-
According to Boxofficemojo.com, the average ticket price in the United States is $8.66. Fifteen years ago in 2001, the average ticket price was $5.66. Prices tend to rise over a 15 year time period...
-
At December 31, 2012 and 2013, Funk & Noble Corporation had outstanding 820 million shares of common stock and 2 million shares of 8%, $100 par value cumulative preferred stock. No dividends were...
-
How do individual companies respond to economic forces throughout the globe? One way to explore this is to see how well rates of return for stock of individual companies can be explained by stock...
-
Calculating Annuity Values Dinero Bank offers you a $30,000, seven-year term loan at 8 percent annual interest. What will your annual loan payment be?
-
Calculating Perpetuity Values The Maybe Pay Life Insurance Co. is trying to sell you an investment policy that will pay you and your heirs $20,000 per year forever. If the required return on this...
-
Calculating Perpetuity Values In the previous problem, suppose a sales associate told you the policy costs $280,000. At what interest rate would this be a fair deal?
-
Wildhorse Corporation, which uses ASPE, enters into a 6-year lease of equipment on September 1, 2023, that requires 6 annual payments of $31,000 each, beginning September 1, 2023. In addition,...
-
12. Consider the following two mutually exclusive investment projects: n Project A Project B 10 -$15,000 -$25,000 1 $5,000 $14,500 2 $8,000 $9,000 3 $5,000 $10,000 $6,000 14 Using the least common...
-
Paula Boothe, president of the Flounder Corporation, has mandated a minimum 9% return on investment for any project undertaken by the company. Given the company's decentralization, Paula leaves all...

Study smarter with the SolutionInn App


