Use the following data to estimate a value of K p at 1200 K for the reaction
Question:
Use the following data to estimate a value of Kp at 1200 K for the reaction 2 H2(g) + O2(g) ⇌ 2 H2O(g)
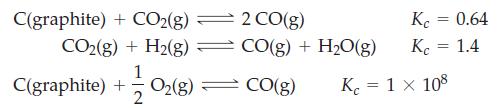
Transcribed Image Text:
C(graphite) + CO2(g) ⇒ CO₂(g) + H₂(g) = C(graphite) + O₂(g) 1 2 2 CO(g) CO(g) + H₂O(g) CO(g) Kc = 0.64 Kc = 1.4 Kc = 1 x 108
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
To estimate the value of Kp at 1200 K for the reaction 2 H2g O2g 2 H...View the full answer

Answered By

Amit Choudhary
I'm new in this profession regarding online teaching but previously i used to teach students near my college. I am teaching on online platform since last year and got good support from the students. I'm teaching on platforms like chegg and vedantu and also at my home in free time.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
The equilibrium constant for the H2 + ½ O2 H2O reaction at 1 atm and 1200 K is KP. Use this information to determine the equilibrium constant for the following reactions: (a) at l atm H, +...
-
Use the following data to estimate ÎHof for magnesium fluoride. Mg(s) + F2(g) MgF2(s) Lattice energy First ionization energy of Mg Second ionization energy of Mg 1445 k/mol Electron affinity of...
-
The current (in amperes) at time t (in seconds) flowing in the circuit in Figure 19 is given by Kirchhoff's Law: i(t) = Cv (t) + R 1v(t) where v(t) is the voltage (in volts), C the capacitance (in...
-
Refer to the information in BE4-7 for Hébert Company. Prepare the correcting journal entries. Information in BE4-7 1. A collection of cash on account from a customer for $750 was recorded as a...
-
What is the operating breakeven point? How do changes in fixed operating costs, the sale price per unit, and the variable operating cost per unit affect it?
-
Thornton fraudulently represented to Daye that he would obtain for her a new car to be used in Dayes business for $17,800 from Pennek Motor Company. Daye thereupon executed her personal check for...
-
The following data are for Marvin Department Store. The account balances (in thousands) are for 2017. 1. Compute (a) the cost of goods purchased and (b) the cost of goods sold. 2. Prepare the income...
-
Grover Corp. is a manufacturing company that produces golf clubs. Birdie is a division of Grover that manufactures putters. Birdies putters are used in Grovers golf club sets and are sold to other...
-
Describe a specific scenario, situation, or application where using a foreign key would be necessary. 2) Explain your reasons, including the characteristics of the data, that necessitate the foreign...
-
Determine K c for the reaction N 2 (g) + O 2 (g) + Cl 2 (g) 2 NOCl(g), given the following data at 298 K. N2(g) + O2(g) NO(g) 102(8) NOCl(g) 1 NO2(g) + Cl(8) NOCl(g) 2 NOCI(g) + Kp = 1.0 10-9 Kp...
-
(A) Excess Ag(s) is added to 1.20 M Fe 3+ (aq). Given that what are the equilibrium concentrations of the species in solution? (B) A solution is prepared with [V 3+ ] = [Cr 2+ ] = 0.0100 M and [V 2+...
-
Find the values of a needed to make the following transformation orthogonal. -a a
-
Should the US pursue an isolationist foreign policy, or should it continue to be active on the global stage? Why?
-
What sectors are driving new recruitments in the UAE?
-
What can a HIPS do that a NIPS cannot? (Choose two.) Detect malware delivered to the host via an encrypted channel. Protect a mobile host while connected to non-secured networks. Block malware as it...
-
Why is it necessary to wear organisation-approved, brightly coloured, waterproof band- aids to cover cuts and abrasions?
-
4. What is the difference in metrics used by the following router protocols: distance vector and link state? 5. How did Classless Inter-Domain Routing (CIDR) help alleviate the problem of too many...
-
Sparkys forecasts that sales will grow by 25% in 2013 and that its cost of goods sold to sales ratio will be the same in 2013 as it was in 2012. Given the information provided about Sparky, what is...
-
we have to compute the letter grades for a course. The data is a collection of student records stored in a file. Each record consists of a name(up to 20 characters), ID (8 characters), the scores of...
-
At the end of June, Lewis Company had completed Jobs 30 and 32. Job 30 is for 1,600 units, and Job 32 is for 1,750 units. Using the data from Practice Exercises 19-1B, 19-2B, and 19-4B, determine (a)...
-
Luek Company completed 60,000 units during the year at a cost of $900,000. The beginning finished goods inventory was 10,000 units at $140,000. Determine the cost of goods sold for 45,000 units,...
-
Suo Company completed 20,000 units during the year at a cost of $120,000. The beginning finished goods inventory was 2,500 units at $14,000. Determine the cost of goods sold for 12,000 units,...
-
An infinitely long wire carries steady current I directly into the page. If the wire goes straight through the center of a sphere of radius R, find the magnetic flux through the sphere.
-
9. Three point charges are oriented as shown below: q = 40 C at the origin and q -20 C on the x-axis at x = 2, and 93 electric force on q2 due the other charges? = = 30 C located at x = 2 cm, y = 2...
-
In (Figure 1), C = C5 = 8.6 F and C = C3 = C = 4.8 F. The applied potential is Vab = 210 V. What is the equivalent capacitance of the network between points a and b? Express your answer in farads....

Study smarter with the SolutionInn App


