Determine K c for the reaction N 2 (g) + O 2 (g) + Cl 2 (g)
Question:
Determine Kc for the reaction N2(g) + O2(g) + Cl2(g) ⇌ 2 NOCl(g), given the following data at 298 K.
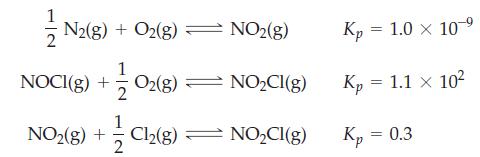
Transcribed Image Text:
N2(g) + O2(g) — NO₂(g) 102(8) NO₂Cl(g) 1 NO2(g) + Cl₂(8) ⇒ NO₂Cl(g) 2 NOCI(g) + Kp = 1.0 × 10-9 Kp = 1.1 × 102 Kp = 0.3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
To determine the equilibrium constant Kc for the given overall reaction N2g O2g Cl2g 2NOCIg Were giv...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
The equilibrium constant for the reaction N2 (g) + O,(g) ;:='02NO(g) is 1.69 x 10-3 at 2300 K. A mixture consisting of 5.0 g of nitrogen and 2.0 g of oxygen in a container of volume 1.0 dm3 is heated...
-
The equilibrium constant Kc for the reaction at 450oC is 0.159. Calculate the equilibrium composition when 1.00 mol N2 is mixed with 3.00 mol H2 in a 5.00-L vessel. N2(g) 3H2(g) 2NH3(g)
-
The molecule methylamine (CH3NH2) can act as a monodentate ligand. The following are equilibrium reactions and the thermochemical data at 298 K for reactions of methylamine and en with Cd2+ (aq); (a)...
-
On March 1, 2014, Eire Co. paid $4,800 to Big North Insurance for a one-year insurance policy. Eire Co. has a December 31 fiscal year end and adjusts accounts annually. Complete the following for...
-
What is operating leverage? What causes it? How is the degree of operating leverage (DOL) measured?
-
Moore issues a negotiable promissory note payable to the order of Parish for the amount of $3,000. Parish raises the amount to $13,000 and negotiates it to Holton for $12,000. (a) If Holton is a...
-
The following data are for Huang Wong Ping Retail Outlet Stores. The account balances (in thousands) are for 2017. 1. Compute (a) the cost of goods purchased and (b) the cost of goods sold. 2....
-
1. Suppose you belong to a tennis club that has a monthly fee of $100 and a charge of $5 per hour to play tennis. a. Using Figure as a model, prepare a table and draw a curve to show the relationship...
-
Why is it important to learn SQL? In which jobs you need to know SQL? 2A- What is the definition of DML and DDL? Give examples in each case. 2B- Consider the following two tables below: VENDOR and...
-
For the reaction 2 H 2 S(g) 2 H 2 (g) + S 2 (g), the equilibrium constant is K c = 4.20 x 10 -6 at 830 C. What are the equilibrium concentrations when 0.500 mol H 2 S is placed in an empty 1.0 L...
-
Use the following data to estimate a value of K p at 1200 K for the reaction 2 H 2 (g) + O 2 (g) 2 H 2 O(g) C(graphite) + CO2(g) CO(g) + H(g) = C(graphite) + O(g) 1 2 2 CO(g) CO(g) + HO(g) CO(g) Kc...
-
Lim is a new pharmacist heavily in debt. A couple of Lims patients at the pharmacy who regularly received prescriptions for controlled substances told Lim they could make it worth his while if he...
-
Grounders Inc. is an American firm that will pay MYR 1,991,000 in a year. The annual interest rates in Malaysia and the US are 10% and 13%, respectively. The spot exchange rate for a US dollar is MYR...
-
Why Trump administration dropped tariffs on Canadian aluminum in September 2020? Did Trump's tariffs benefit American workers and national security?
-
A network is running SNMP on its system. What can be assumed about the network? The network can be accessed remotely. The network cannot be accessed remotely. The network uses *nix OS. The network...
-
Consider this C statement: if (a + b > 30) x = 70; else x= 100; Translate it to ARM assemble language code.
-
What do the activities of the sending computer's presentation layer ensure? The network connection's parameters are agreed on by both the sending and receiving PCs. The physical layer will be able to...
-
The following financial statement data pertains to ABC Company (dollar amounts in millions): Total Assets .........$145,782 Interest-Bearing Debt ...... $32,659 Average Pre-tax borrowing cost......
-
A glass manufacturer produces hand mirrors. Each mirror is supposed to meet company standards for such things as glass thickness, ability to reflect, size of handle, quality of glass, color of...
-
Thomson Company estimates that total factory overhead costs will be $600,000 for the year. Direct labor hours are estimated to be 250,000. For Thomson Company, (a) Determine the predetermined factory...
-
Lewis Company estimates that total factory overhead costs will be $200,000 for the year. Direct labor hours are estimated to be 25,000. For Lewis Company, (a) Determine the predetermined factory...
-
At the end of May, Thomson Company had completed Jobs 70 and 71. Job 70 is for 8,000 units, and Job 71 is for 10,000 units. Using the data from Practice Exercises 19-1A, 19-2A, and 19-4A, determine...
-
The point charges in the figure below are located at the corners of an equilateral triangle 25.0 cm on a side, where qa 9b (a) Find the electric field at the location of qa magnitude direction N/C...
-
Why must a variable be declared with a data type before use? discuss with example
-
1. m A cannonball is fired at an angle of 37.0 above the horizontal and a speed of 33.0- S . It is fired from the top edge of a cliff that is 17.5m high, and land somewhere on the ground below the...
The Condition Of The Working Class In England In 1844 1st Edition - ISBN: 979-8379247737 - Free Book

Study smarter with the SolutionInn App


