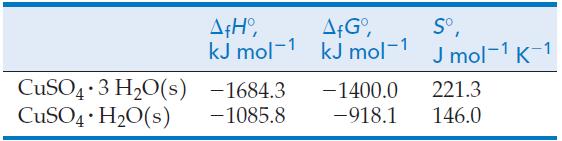
Use the following data together with other data from the text to determine the temperature at which
Question:
Use the following data together with other data from the text to determine the temperature at which the equilibrium pressure of water vapor above the two solids in the following reaction is 75 Torr.
![]()
Transcribed Image Text:
CuSO4 3 H₂O(s) — CuSO4 · H₂O(s) + 2 H₂0(g) . .
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
To determine the temperature at which the equilibrium pressure of water vapor above the two solids in the given reaction is 75 Torr we can use the Van...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Suppose that you have $250 to invest. a). You decide to store the $250 in cash under your mattress. If inflation is 3 percent, what will the real value of that cash be after one year under the...
-
In a study of the reaction 3Fe(s) + 4H2O(g) Fe3O4(s) + 4H2(g) at 1200 K, it was observed that when the equilibrium partial pressure of water vapor is 15.0 torr, the total pressure at equilibrium is...
-
Four vapor pressure data pointstwo representing solidvapor equilibrium and two representing liquidvapor equilibriumare available for a compound: A. Give your best estimate of the triple point...
-
Suppose you needed a material that could absorb heat without having its temperature increase very much. Would you choose aluminum or water? Make sure your explanation uses the concept of specific...
-
In Paige Company, direct labor is $20 per hour. The company expects to operate at 10,000 direct labor hours each month. In January 2014, direct labor totaling $204,000 is incurred in working 10,400...
-
Determine the force in members JI, EF, EI, and JE of the truss, and state if the members are in tension orcompression. 1500 Ib 1000 Ib 1000 Ib 900 lb s'ft DE 8'ft 8'ft 8 ft 8 ft 8 ft' 8 ft'8 ft' 8...
-
Match the measures of worth in the first column with the appropriate unit of measure that results from the analysis. Measure of Worth (a) Annual Worth (b) External Rate of Return (c) Future Worth (d)...
-
Financial information for Ernie Bishop Company is presented below. Additional information: 1. Inventory at the beginning of 2012 was $118,000. 2. Total assets at the beginning of 2012 were $632,000....
-
2. The armadillo is known as one of the world's fastest land animals. On one particular occasion, a biologist observed an armadillo named [N57E] for 6.00 s. The armadillo then ran 19.0 m [N23W]. What...
-
From the data given in Exercise 72, estimate a value of r S at 298 K for the reaction Exercise 72 Sodium carbonate, an important chemical used in the production of glass, is made from sodium...
-
The term thermodynamic stability refers to the sign of r G . If r G is negative, the compound is stable with respect to decomposition into its elements. Use the data in Appendix D to determine...
-
Use nodal analysis to find vo in the circuit of Fig. 10.58. 20 10 mH 10 cos 103, V 2012 4i 302
-
What is the maturity of a default-free security with annual coupon payments and a yield to maturity of 4.6%? Why? Maturity (years) Zero-coupon YTM 1 4.6% 2 5.0% 3 5.4% 4 5.8% 5 6.1%
-
What is the relationship between innovation and economic growth? With reference to specific examples, discuss the effects of intellectual property theft on national income.
-
Refer to the Dodge v. Ford Case. Identify the key arguments for and against the case of abusive managerial discretion. Is this a case that should have been decided in a court of law? Why?
-
What are the two basic rules to follow when computing ES and EF times?
-
Critically review and discuss the scientific evidence in the Daubert v. Merrell Dow Pharmaceuticals Inc. Is expert testimony important? Why? How do judges perform the role of gatekeepers?
-
The following financial statement information is from five separate companies. Required 1. Answer the following questions about Company V: a. What is the amount of equity on December 31, 2012? b....
-
What are the key elements of a system investigation report?
-
Price and efficiency variances, journal entries. The Monroe Corporation manufactures lamps. It has set up the following standards per finished unit for direct materials and direct manufacturing...
-
Continuous improvement the Monroe Corporation sets monthly standard costs using a continuous-improvement approach. In January 2009, the standard direct material cost is $45 par unit and the standard...
-
Materials and manufacturing labor variances, standard costs. Dunn, Inc. is a privately held furniture manufacturer. For August 2009, Dunn had the following standards for one of its products, a wicker...
-
During the first half of a basketball game, a team made 40% of their 30 field goal attempts. During the second half, they scored on only 30% of 40 attempts from the field. What was their field goal...
-
Larner Corporation is a diversified manufacturer of industrial goods. The company's activity-based costing system contains the following six activity cost pools and activity rates: Activity Cost Pool...
-
A furniture store calculates the retail price of furniture they sell by marking up its cost at wholesale a full 100%. (a) If they had a sale with all items marked down 14% from the retail price, what...

Study smarter with the SolutionInn App


