Use the information provided in Figure 21-5 to write balanced chemical equations for the reactions involved in
Question:
Use the information provided in Figure 21-5 to write balanced chemical equations for the reactions involved in synthesizing sodium carbonate from sodium chloride.
Figure 21-5
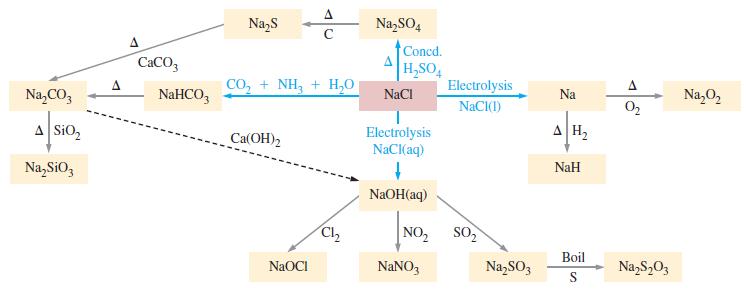
Transcribed Image Text:
Na₂CO3 A SiO₂ Na₂SiO3 A CaCO3 NaHCO3 Na₂S CO, + NH, + H,O Ca(OH) 2 A C NaOCI Cl₂2 Na₂SO4 Concd. H₂SO4 NaCl Electrolysis NaCl(aq) ↓ NaOH(aq) NO₂ NaNO3 Electrolysis NaCl(1) SO₂ Na₂SO3 Na AH₂ NaH Boil S A 0₂ Na₂S₂03 Na₂O₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
Analyze Consult Figure 215 to find a route from NaCl to Na 2 CO 3 One possib...View the full answer

Answered By

William Otieno
I am a professional tutor and a writer with excellent skills that are important in serving the bloggers and other specialties that requires a great writer. The important aspects of being the best are that I have served so many clients with excellence
With excellent skills, I have acquired very many recommendations which have made it possible for me to survive as an excellent and cherished writer. Being an excellent content writer am also a reputable IT writer with essential skills that can make one turn papers into excellent result.
4.70+
83+ Reviews
354+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Iron is extracted from its ores haematite and magnetite. Usually it is extracted from haematite (iron(iii) oxide). The ore is mixed with limestone and coke and reduced to the metal in a blast...
-
Copy and complete the table below, which shows the results of the electrolysis of four substances using inert electrodes. a. State what you understand by inert electrodes. b Explain why the lithium...
-
A 6.53-g sample of a mixture of magnesium carbonate and calcium carbonate is treated with excess hydrochloric acid. The resulting reaction produces 1.72 L of carbon dioxide gas 28oC at and 743 torr...
-
Each table of values gives several points that lie on a line.(a) What is the x-intercept of the line? The y-intercept?(b) Which equation in choices AD corresponds to the given table of values?(c)...
-
You have recently been hired as a staff assistant in the office of the chairman of the board of directors of Clefton, Inc. Because you have some background in accounting, the chairman has asked you...
-
A chairlift has been stopped in the position shown. Knowing that each chair weighs 250 N and that the skier in chair E weighs 765 N, draw the free-body diagrams needed to determine the weight of the...
-
Develop a sales plan, focusing on the organizational structure you would use for your salesforce (geographic, product, or customer).
-
Identify four potential unethical actions or inactions related to capacity planning, and the ethical principle each violates.
-
Which formula in cell C6 would continue to be accurate after a row was inserted between rent expense and insurance expense? 1 Expenses: 2 Wages expense 3 Rent expense 4 Insurance expense 5 Utilities...
-
Use information from the chapter to write chemical equations to represent each of the following: (a) Reaction of cesium metal with chlorine gas; (b) Formation of sodium peroxide (Na 2 O 2 ); (c)...
-
Listed are several pairs of substances. For some pairs, one or both members of the pair react individually with water to produce a gas. For others, neither member of the pair reacts with water. The...
-
Look closely at Burrell and Morgans (1979) four paradigms. Why do you think that there has been almost negligible management research from a radical humanist or radical structuralist perspective?...
-
As an official sponsor of the Olympics, what specific benefit did John Hancock use to help drive sales in their national offices?
-
assumes that Nia has both a discount rate of zero and faces an interest rate of zero. These assumptions made calculating her constant level of consumption expenditure of $56,000 fairly...
-
Paul Petersen lives in Northern California. He owns a BMW car worth about $20,000. He wants to take a trip to Nevada with his girlfriend Patricia, who lives in Los Angeles. He takes his car into...
-
Do you see gendered patterns of interaction in personal relationships? Does knowing about gender linked patterns affect how other interpret on what happens in a relationships?
-
Significance For bone density scores that are normally distributed with a mean of 0 and a standard deviation of 1, find the percentage of scores that are significantly high (or at least 2 standard...
-
In how many ways can 15 (identical) candy bars be distributed among five children so that the youngest gets only one or two of them?
-
Suppose the government bond described in problem 1 above is held for five years and then the savings institution acquiring the bond decides to sell it at a price of $940. Can you figure out the...
-
Does the expected rate of return on the portfolio depend on the percentage of the portfolio invested in each stock? What about the riskiness of the portfolio?
-
Suppose an investor starts with a portfolio consisting of one randomly selected stock. What would happen (1) to the riskiness and to the expected return of the portfolio as more randomly selected...
-
Should the effects of a portfolio impact the way investors think about the riskiness of individual stocks?
-
Which of the following programs covers custodial care? A HMOs B Medicare Part B C PPOs D Medicare Part A E Medicaid
-
uppose a taxpayer has exhausted his lifetime exclusion amount and has $14 million. a. Assuming a flat 40% gift tax rate, what is the maximum amount a taxpayer can transfer to her daughter (and still...
-
Physical Units Method, Relative Sales Value Method Farleigh Petroleum, Inc., is a small company that acquires high - grade crude oil from low - volume production wells owned by individuals and small...
Simple Start How To Get Going With Minimal Investment 1st Edition - ISBN: 979-8389110281 - Free Book

Study smarter with the SolutionInn App


