Use the value of the van der Waals constant b for He(g) given in Table 6.5, to
Question:
Use the value of the van der Waals constant b for He(g) given in Table 6.5, to estimate the radius, r, of a single helium atom. Give your answer in picometers.
Table 6.5
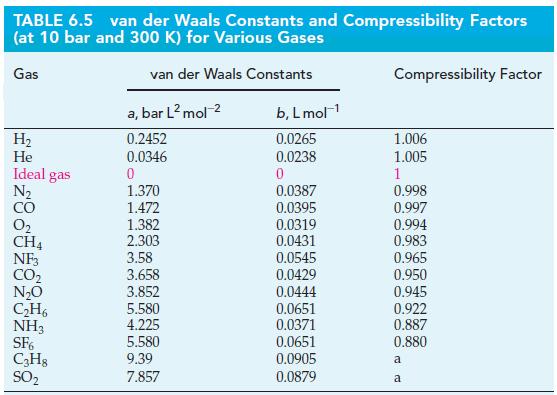
Transcribed Image Text:
TABLE 6.5 van der Waals Constants and Compressibility Factors (at 10 bar and 300 K) for Various Gases van der Waals Constants Gas H₂ He Ideal gas N₂ CO 0₂ CH4 NF3 CO₂ N₂O C₂H6 NH3 SF6 C3H8 SO₂ a, bar L² mol-² 0.2452 0.0346 0 1.370 1.472 1.382 2.303 3.58 3.658 3.852 5.580 4.225 5.580 9.39 7.857 b, L mol-¹ 0.0265 0.0238 0 0.0387 0.0395 0.0319 0.0431 0.0545 0.0429 0.0444 0.0651 0.0371 0.0651 0.0905 0.0879 Compressibility Factor 1.006 1.005 1 0.998 0.997 0.994 0.983 0.965 0.950 0.945 0.922 0.887 0.880 a a
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
According to Table 65 the van der Waals constant b for Heg is 00238 Lmol We can use this va...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
(a) Use the value of the van der Waals constant b for CH 4 (g), given in Table 6.5, to estimate the radius of the CH 4 molecule. (See Exercise 89.) How does your estimate of the radius compare with...
-
A certain gas obeys the van der Waals equation with a =0.76 m6 Pa mol-2, its volume is found to be 4.00 X 10-4 m3 mol-1 at 288 K and 4.0 MPa. From this information calculate the van der Waals...
-
The volume of a spherical molecule can be estimated as V = b/(4N A ), where b is the van der Waals parameter for the excluded molar volume and N A is Avogadros number. Justify this relationship by...
-
In Problems, assume that the populations are normally distributed and that independent sampling occurred. (a) Test the hypothesis that 1 2 at the a = 0.1 level of significance for the given sample...
-
Prepare a WBS using MSP with the steps that must be completed before Vern Toney can contact outsourcing vendors. If Vern starts on August 1, 2005, how long will it take to get ready to contact...
-
Assume the same information in E16-1, except that Angela Corporation converts its convertible bonds on January 1, 2016. Instructions (a) Compute the carrying value of the bond payable on January 1,...
-
Hooters Restaurant in Myrtle Beach, South Carolina, used an alternative dispute resolution program, a program to resolve disputes outside the traditional court system. Employees of Hooters had to...
-
Ohno Company specializes in manufacturing a unique model of bicycle helmet. The model is well accepted by consumers, and the company has enough orders to keep the factory production at 10,000 helmets...
-
8. Draw and label a potential graph and contour plot for: a. A point charge of 1nC. b. A parallel-plate capacitor with AV = 2.0V 9. The electric potential generated by a particular point charge is...
-
Refer to Example 6-17. Recalculate the pressure of Cl 2 (g) by using both the ideal gas equation and the van der Waals equation at the temperatures (a) 100 C; (b) 200 C; (c) 400 C. From the results,...
-
Use both the ideal gas equation and the van der Waals equation to calculate the pressure exerted by 1.50 mol of SO 2 (g) when it is confined at 298 K to a volume of (a) 100.0 L; (b) 50.0 L; (c) 20.0...
-
On October 1, 2013, Koppa Corp. issued $700,000, 5%, 10-year bonds at face value. The bonds were dated October 1, 2013, and pay interest annually on October 1. Financial statements are prepared...
-
Let A = 41^-7^ B = 61-1j^ C = -91 + 5J^ Complete each vector sum. A+B+C = A-B+C = + + A+B-C= A - B - C = + +
-
Northern Warrior Cooperative Berhad (NWCB) has been in the operation for the last 10 years. For the year ended 30 September 2023, the following information were extracted from the audited accounts as...
-
You review a company's financial statements and it reveals that its inventory days figure is erratic, ranging from 20 days to 75 days. The company's inventory is not likely to become obsolete and is...
-
3. Four mutual friends are avid texters: at any time, each friend has a probability Xh+o(h) of sending a text message in the next time interval of length h. Whenever any friend sends a message, it is...
-
2. A linear system with no free variables: Consider the linear system x1 + 2x2 + 3x3 2x1 - x2 + 3x3 4x1 - x2 + 12x3 You'll solve this linear system in a few ways: = 115 = 1421 = 4214 (a) First, enter...
-
Find the Consolidated Statement of Stockholders Equity and Comprehensive Income section of the Home Depot 2009 financial statements in Appendix A. Locate the translation adjustment for 2009. Was the...
-
Les has collected stamps in his spare time for years. He purchased many of his stamps at a price much lower than the current market value. Les recently lost his job as a carpenter. Since his wife...
-
What are the general rules for measuring gain or loss by both creditor and debtor in a troubled-debt restructuring involving a settlement?
-
(a) In a troubled-debt situation, why might the creditor grant concessions to the debtor? (b) What type of concessions might a creditor grant the debtor in a troubled-debt situation?
-
What are the general rules for measuring and recognizing gain or loss by both the debtor and the creditor in a troubled-debt restructuring involving a modification of terms?
-
Karisma Global Berhad has been a public listed company with Bursa Malaysia since 2012. The principal activity of the company is manufacturing fabricated aluminum valve which is mainly exported to...
-
BP, a UK energy company, has just signed a contract on 1st June 2020 to import $5,500,000 of raw materials from a US company. The payment is due in six months' time. The board of BP expect a steep...
-
discuss Digital Transformation impact on marketing research.

Study smarter with the SolutionInn App


