Which of the following reactions are likely to go to completion or very nearly so? (a) (b)
Question:
Which of the following reactions are likely to go to completion or very nearly so?
(a)![]()
(b)
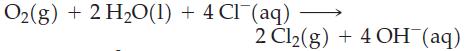
(c)
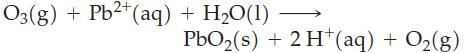
(d)
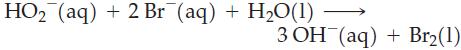
Transcribed Image Text:
H₂O₂(aq) + 21¯(aq) + 2 H¹(aq) I2(s) + 2 H₂O(1)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
To determine which reactions are likely to go to completion or very nearly so we can analyze the rea...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Homework-2 DECISION ANALYSIS PROBLEMS Problem-1: Kenneth Brown is the principal owner of Brown Oil, Inc. After quitting his university teaching job, Ken has been able to increase his annual salary by...
-
Project the 2 4-1 IV design in Example 8-1 into two replicates of a 2 2 design in the factors A and B. Analyze the data and thaw conclusions. Example 8-1: Consider the filtration rate experiment in...
-
KYC's stock price can go up by 15 percent every year, or down by 10 percent. Both outcomes are equally likely. The risk free rate is 5 percent, and the current stock price of KYC is 100. (a) Price a...
-
CPA firm brings in a yoga instructor during the tax busy season to help relieve stress of the employees. Which is true about the CPA firm's ability to take a deduction for the yoga instructor's...
-
Refer to the information in E 8-24. If those holding stock options can purchase a share of stock for $48 and the market value of a share of stock on January 1, 2012, is $50, how can the option to...
-
What procedures, if any, do professional standards require auditors to perform when reviewingprior to the completion of the audita clients post-year-end earnings press release?
-
Estimate the overall odds ratio of the set of tables in Problem3.6 and test whether the odds ratios are the same across the tables. Problem3.6 is: 3.6 Use the DOS data to test whether there is gender...
-
Mohave Corp. is considering eliminating a product from its Sand Trap line of beach umbrellas. This collection is aimed at people who spend time on the beach or have an outdoor patio near the beach....
-
How to create new user ASSIGN-1 in Oracle database and get login with it. User must not be given DBA rights. give permission to ASSIGN-1 to create and view tables.
-
Use Lewis structures and other information to explain the observation that (a) H 2 S is a gas at room temperature, whereas H 2 O is a liquid. (b) O 3 is diamagnetic.
-
In water, O 2 (aq) is a strong base. If 100.0 mg of K 2 O(s) is dissolved in 1.25 L of aqueous solution, what will be the pH of the solution?
-
Describe the inputs needed for capacity requirements planning.
-
(Fill in each step after checking the law of your state, particularly your state statutory code. You may also need to refer to court rules and judicial opinions. See General Instructions for the...
-
Explain the two methods to account for stock investments in companies that own less than 50% of the voting stock.
-
The CEO of Sunway Corporation is puzzled. During the year, the company experienced a loss of $800,000, yet its cash increased $300,000 during the same period. Explain how this situation occurred.
-
Write an individual or a team report. Pick one of the following topics. 1. Improving Customer Service. Many customers find that service is getting poorer and workers are getting ruder. Evaluate the...
-
The following opening and closing lines come from class presentations about informational interviews. Assess: How well does each opening create interest in the rest of the presentation? (How well...
-
Tom is a CPA for a large regional firm. In preparing the tax return for Espresso Industries, he notices that the firm has an unusually high amount of travel, meal, and entertainment expenses....
-
The bookkeeper for Riley, Inc., made the following errors: a. A cash purchase of supplies of $357 was recorded as a debit to Supplies for $375 and a credit to Cash of $375. b. A cash sale of $3,154...
-
What principles of internal control apply to most businesses?
-
Graham Moran is reviewing the principle of segregation of duties. What are the two common applications of this principle?
-
How do documentation procedures contribute to good internal control?
-
What are the quality measures in EHR implementation in a hospital setting/LTC? What are the steps for developing, monitoring, and measuring a quality improvement process for a strategic plan of an...
-
A barber shop uses process capability to measure the consistency in service times for different customers. The average service time is 40 minutes with a standard deviation of 10 minutes. To meet...
-
Determine the cubic feet of mortar and number of 8x8x16 inch concrete blocks required to construct a building's foundation (4 sides) that are 42 ft wide, 60 ft long and 10 ft high. Assume a 10% waste...

Study smarter with the SolutionInn App


