Will reaction (23.25) still be spontaneous in the forward direction in a solution containing equal concentrations of
Question:
Will reaction (23.25) still be spontaneous in the forward direction in a solution containing equal concentrations of Fe2+ and Fe3+, a pH of 3.25, and under an O2(g) partial pressure of 0.20 atm?
Reaction (23.25)
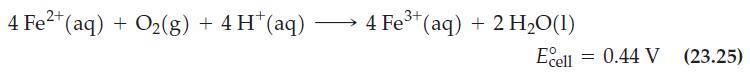
Transcribed Image Text:
4 Fe²+ (aq) + O₂(g) + 4H+ (aq) 3+ → 4 Fe³+ (aq) + 2 H₂O(1) Ecell = 0.44 V (23.25)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
The spontaneity of a reaction can be determined by the Gibbs Free Energy AG of the reaction The relationship between AG standard cell potential Ecell ...View the full answer

Answered By

Zablon Gicharu
I am an educator who possesses the requisite skills and knowledge due to interacting with students for an extended period. I provide solutions to various problems in step-by-step explanations, a well-thought approach and an understandable breakdown. My goal is to impart more straightforward methodologies and understanding to students for more remarkable achievements.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
The reaction coordinate diagram in Figure 15.5 shows that the rate-determining step for sulfonation is the slower of the two steps, whereas the rate-determining step for desulfonation is the faster...
-
The following r G values are given for 25 C. Combine the preceding equations, as necessary, to obtain r G values for the following reactions. Of reactions (a), (b), and (c), which is spontaneous in...
-
1. Determine the factory overhead rates under the multiple production department rate method. Assume that indirect labor is associated with the production departments, so that the total factory...
-
How could not having a strategy hurt a brand on social media? What are some content ideas for those who are at the "Awareness" stage of the sales funnel? What does it mean to A/B test on a social...
-
As a consultant for ABC Consulting Company, you have been hired by Eddies Electronics, a company that owns 25 electronics stores selling radios, televisions, compact disc players, stereos, and other...
-
The CPA firm of May & Marty has audited the consolidated financial statements of BGFI Corporation, a private company. May & Marty examined the parent company and all subsidiaries except for BGI-...
-
Dr. Walter Sullivan was one of several plastic surgeons in Las Vegas visited by Julie Jones. Jones, an exotic dancer, sought plastic surgery to improve her ability to make money in her profession....
-
Eight Flags operates several amusement parks in the Midwest. The company stocks machine oil to service the machinery for the many rides at the parks. Eight Flags needs 30,000 gallons of oil annually;...
-
Part A An oxygen atom at a particular site within a DNA molecule can be made to execute simple harmonic motion when illuminated by infrared light. The oxygen atom is bound with a spring-like chemical...
-
What products are obtained when Mg 2+ (aq) and Cr 3+ (aq) are each treated with a limited amount of NaOH(aq)? With an excess of NaOH(aq)? Why are the results different in these two cases?
-
Why is it reasonable to expect the chemistry of dichromate ion to involve mainly oxidationreduction reactions and that of chromate ion to involve mainly precipitation reactions?
-
How does depreciation affect the cash balance?
-
One difference between accounting for a governmental (not-for-profit) unit and a commercial (for-profit) enterprise is that a governmental unit should a. Not record depreciation expense in any of its...
-
The Euro has become a reserve currency held by foreign central banks. Suppose that some of these banks held Euro reserves in the form of Euro notes kept in their vaults rather than in the form of...
-
Jim Watanabe, assistant director of IT for Petrie Electronics, and the manager of the No Customer Escapes customer loyalty system project, was walking down the hall from his office to the cafeteria....
-
According to GASB 34, infrastructure fixed assets a. Must be capitalized and depreciated. b. Must be capitalized, but governments do not have to depreciate them. c. May be capitalized and...
-
Determine the annual net cost of these checking accounts: a. Monthly fee $5, check-processing fee of 25 cents, average of 19 checks written per month b. Annual interest of 2.5% paid if balance...
-
Lian is injured in an automobile accident this year. She is hospitalized for 4 weeks and misses 3 months of work after getting out of the hospital. The costs related to her accident are...
-
Draw a Feynman diagram for the reaction n + v p + .
-
Canney Company borrows $90,000 on July 1 from the bank by signing a $90,000, 7%, 1-year note payable. Prepare the journal entries to record (a) The proceeds of the note and (b) Accrued interest at...
-
Home Town Supply does not segregate sales and sales taxes at the time of sale. The register total for March 16 is $10,388. All sales are subject to a 6% sales tax. Compute sales taxes payable and...
-
Franklin University sells 3,500 season basketball tickets at $80 each for its 10-game home schedule. Give the entry to record (a) The sale of the season tickets and (b) The revenue earned by playing...
-
Microsoft Access is a database management system. You may learn about MS Access basics on your own: creating tables and running queries. How useful is a database management system to an accountant?...
-
2. You are designing a part that will be made from a commercial grade steel. They give you the following data for stock steel: Yield Strength (MPa) 420 Grain Size (mm) .05 470 550 .024 0.015 Your...
-
P5 (15 Pts): The impeller of a radial water pump has an outer diameter of 10 in and rotates at 1600 rpm. A water flow of 1200 gpm enters the impeller axially and leaves at an absolute velocity of 90...

Study smarter with the SolutionInn App


