Will the following precipitates form under the given conditions? (a) Pbl(s), from a solution that is 1.05
Question:
Will the following precipitates form under the given conditions?
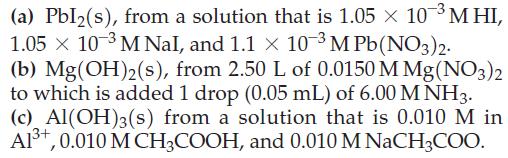
Transcribed Image Text:
(a) Pbl₂(s), from a solution that is 1.05 × 10-³ MHI, 1.05 × 10-3 M Nal, and 1.1 x 10-3 M Pb(NO3)2. (b) Mg(OH)2(s), from 2.50 L of 0.0150 M Mg(NO3)2 to which is added 1 drop (0.05 mL) of 6.00 MNH3. (c) Al(OH)3(s) from a solution that is 0.010 M in Al³+, 0.010 M CH3COOH, and 0.010 M NaCH3COO.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Precipitate Conditions Will Precipitate PbI2s 1...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Marx and Engels were two of the most important philosophers in history. Their influence is widely spread around the world to this day. Marx and Engels are deeply critical of the system of life that...
-
Case Study: Quick Fix Dental Practice Technology requirements Application must be built using Visual Studio 2019 or Visual Studio 2017, professional or enterprise. The community edition is not...
-
a) which ionization gives the best sensitivity the analysis of chloramphenicol? b) The compound with highest retention factor? c) What is the resolution between yohimbine and chloramphenicol? d) What...
-
Taxable income and income tax rates for 20092011 for the company have been as follows: Make the journal entry necessary to record any net operating loss (NOL) carryback in2011. Taxable Income Income...
-
Use Holt-Winters multiplicative method to create a seasonal model that minimizes the MSE for the data set. Use Solver to determine the optimal values of , , and . a. What are the optimal values of ,...
-
In 2004, Jui-Chen Lin, a citizen of China, entered into an agreement with Robert Chiu and Charles Cobb, citizens of the United States, to form an LLC to acquire and operate a fast-food restaurant in...
-
1. Help Jenny to forecast dividend payments for Reeby Sports and to estimate the value of the stock. You do not need to provide a single figure. For example, you may wish to calculate two figures,...
-
Why would you perform a silent installation? What is the purpose of a password complexity policy? Oracle offers a free download of all editions of the Oracle Database. How does the company derive...
-
PbCl 2 (s)is considerably more soluble in HCl(aq) than in pure water, but its solubility in HNO 3 (aq) is not much different from what it is in water. Explain this difference in behavior.
-
To 0.350 L of 0.150 M NH 3 is added 0.150 L of 0.100 M MgCl 2 . How many grams of (NH 4 ) 2 SO 4 should be present to prevent precipitation of Mg(OH) 2 (s)?
-
XYX Inc is evaluating a project. XYX intends to use 20% debt in funding the project. XYX identifies 5 comparable companies with the following equity betas and debt/equity ratios. While the comparable...
-
If an individual owns only one security the most appropriate measure of risk is a.correlation. b.the risk-free rate. c.beta. d.covariance. e.standard deviation.
-
Banz and Reinganum found that small firms consistently outperformed large firms. This anomaly is referred to as the a.growth firm effect. b.small firm effect. c.size effect. d.large firm...
-
2. Consider the following two premises P1, P2, and P3: P: Vx(A(x) B(x)) P2: x(C(x) AB(x) P3: Vx(C(x)(A(x) V D(x)) Prove the following logical implication P1, P2, P3 = 3x C(x) ^ y D(y).
-
1.Discuss the purposes and scope of temporary regulations. 2.Are letter rulings of precedential value to third parties? 3. In 1998, Congress passed legislation concerning shifting the burden of proof...
-
Why is not for profit business a fit organizational structure for a starting up behavioral health clinic? What is the financial manager's role and identify how the finance department fits into the...
-
Refer to the situation described in BE 10-4. What is the carrying value of the asset retirement liability at the end of one year? Assuming that the actual restoration costs incurred after extraction...
-
A company pledges their receivables so they may Multiple Choice Charge a factoring fee. Increase sales. Recognize a sale. Collect a pledge fee. Borrow money. Failure by a promissory notes' maker to...
-
Kwun, Inc. purchases 1,000 shares of its own previously issued $5 par common stock for $12,000. Assuming the shares are held in the treasury, what effect does this transaction have on (a) Net income,...
-
The treasury stock purchased in question 14 is resold by Kwun, Inc. for $18,000.What effect does this transaction have on (a) Net income, (b) Total assets, (c) Total paid-in capital, and (d) Total...
-
(a) What are the principal differences between common stock and preferred stock? (b) Preferred stock may be cumulative. Discuss this feature. (c) How are dividends in arrears presented in the...
-
2. Determine the angles Theta and Phi for the system of weights and pulleys shown below. Assume that the system is in equilibrium. What happens to the angles if pulley A is raised to the same height...
-
A 16-100th pinion drives the double-reduction helical-gear train in the figure. The transmitted power is 5 hp. All gears have 25 normal pressure angle and 20 as helix angle. Pinion 2 rotates cow at...
-
Calculate the beta of security D with the S&P. Year A, % B, % C, % D, % E, % 1 S&P, % 10.67-3.76 12.98 25.51 8.96 0.34 2 12.54 21.67 45.23 18.97 29.67 15.94 3 -8.82 19.84 16.52 -11.57-12.07 4.66 4...

Study smarter with the SolutionInn App


