Without performing detailed calculations, indicate whether any of the following reactions would occur to a measurable extent
Question:
Without performing detailed calculations, indicate whether any of the following reactions would occur to a measurable extent at 298 K.
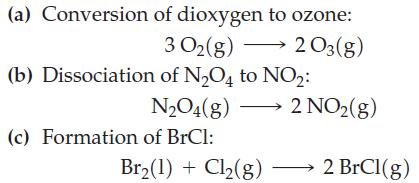
Transcribed Image Text:
(a) Conversion of dioxygen to ozone: 3 0₂(g) → 2 03(g) (b) Dissociation of N₂O4 to NO₂: N₂O4(g) - (c) Formation of BrCl: Br₂(1) + Cl₂(g) 2 NO₂(g) 2 BrCl(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
To determine whether a reaction would occur to a measurable extent at 298 K we can consider the spon...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Which of the following reactions would occur to a measurableextent in water? Br - (aq) + H 2 O (l) ---> HBr (aq) +OH - (aq) NH 4 + (aq) + H 2 O (l) --->NH 3 (aq) + H 3 O + (aq) Cl - (aq) + H 2...
-
Use data from Table 10.3, but without performing detailed calculations, determine whether each of the following reactions is exothermic or endothermic. Table 10.3 (a) CH4(g) + I(g) (b) H(g) + I2(g)...
-
A mover lifts a 50 lbm box off the ground and places it on a truck (Figure 1-21). If the floor of the truck is 4 feet off the ground, how much work was required to lift the box? 4 ft 50 lbs...
-
Venetian Company has two production departments, Fabricating and Assembling. At a department managers meeting, the controller uses flexible budget graphs to explain total budgeted costs. Separate...
-
The jib crane is supported by a pin at C and rod AB. The rod can withstand a maximum tension of 40 kN. If the load has a mass of 2 Mg, with its center of mass located at G, determine its maximum...
-
Consider the following cash flow profile and assume MARR is 10 percent/year. a. What does Descartes' rule of signs tell us about the IRR(s) of this project? b. What does Norstrom's criterion tell us...
-
Profits have been decreasing for several years at Pegasus Airlines. In an effort to improve the companys performance, consideration is being given to dropping several flights that appear to be...
-
Crystal Corporation earned net income of $ 9 0 0 , 0 0 0 in 2 0 2 3 . It has a complex capital structure as follows: 1 2 , 0 0 0 shares outstanding of 8 % , $ 1 0 0 preferred stock, and 1 7 0 , 0...
-
Two correct statements about the reversible reaction N 2 (g) + O 2 (g) 2 NO(g) are (a) K = K p ; (b) The equilibrium amount of NO increases with an increased total gas pressure; (c) The equilibrium...
-
If r G = 0 for a reaction, it must also be true that (a) K = 0; (b) K = 1; (c) r H = 0; (d) r S = 0; (e) The equilibrium activities of the reactants and products do not depend on the initial...
-
Assume that you just won the state lottery. Your prize can be taken either in the form of $40,000 at the end of each of the next 25 years (that is, $1,000,000 over 25 years) or as a single amount of...
-
List each Airbus aircraft type and the competing Boeing type.
-
What is the difference between borrowing funds from a bank for fleet expansion and leasing aircraft?
-
What did the CAB regulate? Why?
-
How do aircraft direct operating costs vary with aircraft size, segment length, and age?
-
Marian Plunket owns her own business and is considering an investment. If she undertakes the investment, it will pay $40,000 at the end of each of the next three years. The opportunity requires an...
-
The ledger of Mai Company includes the following accounts with normal balances: D. Mai, Capital $9,000; D. Mai, Withdrawals $800; Services Revenue $13,000; Wages Expense $8,400; and Rent Expense...
-
Use the graphs of f and g to graph h(x) = (f + g) (x). To print an enlarged copy of the graph, go to MathGraphs.com. 1. 2. y 24 8. 2. -2 -2 4 6
-
The farmer does not avoid risk by selling wheat futures. If wheat prices stay about $2.80 a bushel, then he will actually have lost by selling wheat futures at $2.50. Is this a fair comment?
-
Explain the chief differences between futures and forward contracts, e.g., for foreign exchange.
-
List some of the commodity futures contracts that are traded on exchanges. Who do you think could usefully reduce risk by buying each of these contracts? Who do you think might wish to sell each...
-
Pension funds pay lifetime annuities to recipients. If a firm remains in business indefi- nitely, the pension obligation will resemble a perpetuity. Suppose, therefore, that you are managing a...
-
I'm not going to buy from that store down the street, they don't have the quality of products that I want for my family. I will drive across the tracks (a different community) to buy want I want. "...
-
The local supermarket normally sells potato chips for $3 a packet and sells 100 packets per week. If the supermarket then discounts the potato chips to $2 a packet and sells 160 packets per week....

Study smarter with the SolutionInn App


