Write a balanced chemical equation for the reaction depicted below. +
Question:
Write a balanced chemical equation for the reaction depicted below.
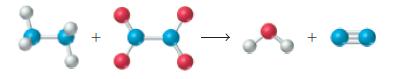
Transcribed Image Text:
+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
The balanced chemical equation for the reaction depicted in the image is 2 SiCl4 2 NH4Cl 2 SiF4 2 N...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Write a balanced chemical equation for the reaction that occurs when (a) Calcium metal undergoes a combination reaction with O2(g) (b) Copper(II) hydroxide decomposes into copper(II) oxide and water...
-
Write a balanced chemical equation for the reaction that occurs when (a) Mg(s) reacts with Cl2(g) (b) Barium carbonate decomposes into barium oxide and carbon dioxide gas when heated (c) The...
-
Write a balanced chemical equation for the pentose phosphate pathway in the first two modes depicted in Figure 12.36, where (a) ribose-5-phosphate synthesis is maximized (b) NADPH production is...
-
A company is deciding whether to produce a new gadget at a plant located in a country close to consumers at a higher labor cost and shorter lead time or to outsource it to a country with a low labor...
-
Using the data for Geo-Metrics Corporation in Exercise 14-20, assume that as of December 31, 2008, the M-Labs Inc. stock had a market value of $25 per share and the Spectrum Corp. stock had a market...
-
What are the possible causes of (a) Wage rate (b) Labour efficiency variances?
-
Air at \(25^{\circ} \mathrm{C}\) flows normal to the axis of an infinitely long cylinder of \(1.0-\mathrm{m}\) radius. The cylinder is rotating at \(10 \mathrm{rad} / \mathrm{s}\), and the approach...
-
Selected account balances and transactions of Alpine Manufacturing Co. follow: May Transactions: a. Purchased raw materials and factory supplies on account at costs of $45,000 and $10,000,...
-
Use logarithmic differentiation to find the derivative of y with respect to the given independent variable. y = (sin 8x)x
-
Phosphorus trichloride, PCl 3 is a commercially important compound used in the manufacture of pesticides, gasoline additives, and a number of other products. A ball-and-stick model of PCl 3 is shown...
-
Write balanced equations to represent: (a) The reaction of sulfur dioxide gas with oxygen gas to produce sulfur trioxide gas (one of the reactions involved in the industrial preparation of sulfuric...
-
Repeat the requirements of P8-9 assuming Wayne Computer Consultants uses the completed-contract method to report its long-term contracts. Data from P8-9 R. Wayne Computer Consultants, Incorporated...
-
20 xn + f(x) = [(1)n+1, n=1 Write a C program that computes the value of the above math function for a given x using the concept of functions in C. Your program should test your C function double...
-
Describe the use and development of GIS within the context of vulnerability assessment
-
The story of Canada's trade relationship with China presents several challenges related to international trade law and the topics of the course. Some of the challenges include: Resolution of dispute:...
-
Simplify V x12 9
-
What is meant by "informed consent"? When might it be appropriate for a company to use personality tests or drug tests in determining who to hire?
-
In a simple linear regression based on 25 observations, it is found that b1 = 0.5 and se(bj) = 0.3. Consider the hypotheses: H0: 1 0 and HA: 1 > 0. a. At the 5% significance level, find the critical...
-
QUESTION 9 HC-O-C-R R-C-O-CH HC-O-P-O-CH-CH-NH3* O || O a. Phosphatidic acid, Serine O b. Lysophosphatidic acid, Serine, Free FA O c. Lysophosphatidylserine, Free FA O d. 2 Free FAs, Serine, Glycerol...
-
What is free cash flow? Why is it the most important measure of cash flow?
-
If you were starting a business, what tax considerations might cause you to prefer to set it up as a proprietorship or a partnership rather than as a corporation?
-
An investor recently purchased a corporate bond which yields 9 percent. The investor is in the 36 percent combined federal and state tax bracket. What is the bonds after-tax yield?
-
The components of vectors A and B are given as follows: Ax=-4 Bx = 17 =- Ay=-10 By = - 3 What is the angle in degrees between the vector difference BA and the positive x-axis?
-
Cullumber Corporation has the following information available about its July, 2 0 2 2 production: Finished goods inventory on July 1 , 2 0 2 2 is $ 2 7 , 1 6 0 and on July 3 1 , 2 0 2 2 is $ 2 4 , 7...
-
B. Berjasa bhd (BB) uses a standard costing system in their production of steel products. Since bB are more automated based production, machine hour is used as the basis in production. At the...

Study smarter with the SolutionInn App


