Write the structures of the isomers you would expect to obtain in the mononitration of m-methoxybenzaldehyde: CHO
Question:
Write the structures of the isomers you would expect to obtain in the mononitration of m-methoxybenzaldehyde:
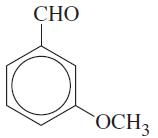
Transcribed Image Text:
CHO OCH₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
CHO OCH3 mMethoxybenzaldehyde 3Methyoxybenzeneald...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
You have synthesized a pure molecule with molecular formula SOF4. However, you remember that it has two possible isomers ) and are trying to determine which isomer you have synthesized. Both isomers...
-
Draw structures for (he products you would expect to obtain from reaction of -D-talopyranose with each of the following reagents: (a) NaBH 4 in H 2 O (b) Warm dilute HNO 3 (c) Br 2 , H 2 O (d) CH 3...
-
Ninety kilograms of sodium nitrate is dissolved in 110 kg of water. When the dissolution is complete (at time r = 0), pure water is fed to the tank at a constant rate m (kg/min), and solution is...
-
Though the McDonalds (MCD) menu of hamburgers, cheeseburgers, the Big Mac, Quarter Pounder, Filet-O-Fish, and Chicken McNuggets is easily recognized, McDonalds financial statements may not be as...
-
Give two examples of facility support activities.
-
In 2011, eBay had $11.6 billion in revenue and net income over $3.2 billion. eBays mission is to provide a global trading platform where practically anyone can trade practically anything. However,...
-
Unless otherwise specified, which rule will always be processed?
-
Staircase Equipment Company uses a job order cost system. The following data summarize the operations related to production for April 2010, the first month of operations: a. Materials purchased on...
-
A trebuchet is a mighty siege engine capable of flinging 90-kg projectiles a distance of 300 meters. Assuming a perfect launch angle of 45, calculate the speed with which one of these projectiles...
-
In the chlorination of CH 4 , some CH 3 CH 2 Cl is obtained as a product. Explain why this should be so.
-
Match the following compounds with the chemical properties given below. Write the structure of the products of the reactions described in (a) to (e). (a) Neutralizes HCl(aq); (b) Neutralizes...
-
Refer back to question 17. Suppose that the nearest index futures contract matures in seven months rather than in six. Show how Legs Diamond can still use index futures to hedge his position. How...
-
2. For each situation below, an object is at rest with a net force of zero. All forces are shown. Calculate and label the missing force magnitude. C. a. F= F=5N F = 20 N F=5N F = 20 N F=5N 1 b. d. F=...
-
What is the electric field (in N/C) at a point where the force on a -3.0 x 10-6 C charge is (4.51-6.6) x 10-6 N? (Express your answer in vector form.) E = t N/C Additional Materials Reading
-
Supergirl is doing a dead lift. She lifts a 10-kg tanuki from the floor to just below her waist (a vertical distance of 0.50 m) in 1 s. What is Supergirl's power in this process? (Assume g to be 10...
-
A flywheel is made from a circular metal disc of mass 50 kg and radius 0.3 m, with a rotation axle through the centre perpendicular to the disc plane. When the flywheel is rotating at a rate of 2000...
-
A reaction involving the gain of one or more electrons is a(n) reaction. O inclusion, O elimination O reduction O oxidation
-
The City of Los Angeles provided equal monthly retirement benefits for men and women of the same age, seniority, and salary. The benefits were partially paid for by employee contribution and...
-
A copper sphere of 10-mm diameter, initially at a prescribed elevated temperature T;, is quenched in a saturated (1 atm) water bath. Using the lumped capacitance method, estimate the time for the...
-
Schweser Satellites Inc. produces satellite earth stations that sell for $100,000 each. The firms fixed costs, F, are $2 million, 50 earth stations are produced and sold each year, profits total...
-
Now calculate the corporate value. Assume you have just been hired as a business manager of PizzaPalace, a regional pizza restaurant chain. The companys EBIT was $50 million last year and is not...
-
Pettit Printing Company has a total market value of $100 million, consisting of 1 million shares selling for $50 per share and $50 million of 10% perpetual bonds now selling at par. The companys EBIT...
-
Using the information in the tables single-factor model. please calculate the expected excess return for this security using a Claythorne International Rogers and Rogers Capital Wargrave Limited...
-
For the following loan, make a table showing the amount of each monthly payment that goes toward principal and interest for the first three months of the loan. A home mortgage of $ 1 4 8 comma 0 0 0...
-
Given the information below, what is the cross-rate of the US dollar in the Japanese yen (i.e., JPY/USD)? Japanese yen in euros (EUR/JPY) US dollar in euros (EUR/USD) US dollar in Japanese yens...

Study smarter with the SolutionInn App


