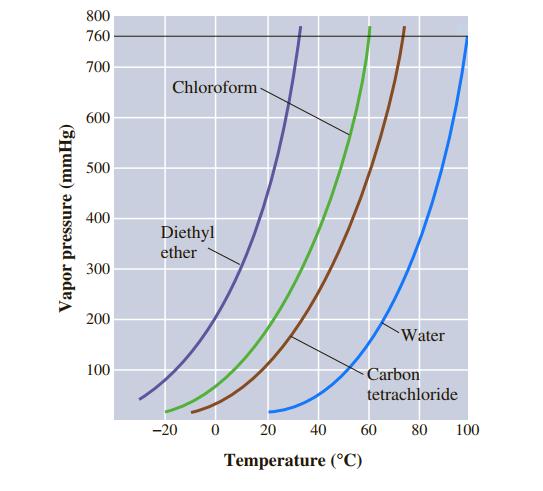
Use Figure 11.7 to estimate the boiling point of diethyl ether, (C 2 H 5 ) 2
Question:
Use Figure 11.7 to estimate the boiling point of diethyl ether, (C2H5)2O, under an external pressure of 470 mmHg.
Transcribed Image Text:
800 760 700 Chloroform- 600 500 400 Diethyl ether 300 200 Water 100 Carbon tetrachloride -20 20 40 60 80 100 Temperature (°C) Vapor pressure (mmHg)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (14 reviews)
Dropping a line from the int...View the full answer

Answered By

Seema kuldeep
although I don't have an experience of teaching in a particular institute, previously I was an expert on Chegg and I have used to teach my batch mates and also my juniors.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Using the vapor-pressure curves in Figure 11.25, (a) Estimate the boiling point of ethanol at an external pressure of 200 torr; (b) Estimate the external pressure at which ethanol will boil at 60 oC;...
-
Use Figure 10.8 to estimate the boiling point of ethanol at 400 torr.
-
Use Figure 11.7 to estimate the boiling point of carbon tetrachloride, CCl 4, under an external pressure of 250 mmHg. 800 760 700 Chloroform- 600 500 400 Diethyl ether 300 200 Water 100 Carbon...
-
a. Find the nth-order Taylor polynomials for the given function centered at the given point a, for n = 0, 1, and 2.b. Graph the Taylor polynomials and the function. f(x)=x, a = 9
-
An investment manager expects a stock to be quite volatile and is considering the purchase of either a straddle or a chooser option. The stock is priced at 44, the exercise price is 40, the...
-
The body of a method is enclosed in __________. a. Curly braces { } b. Square brackets [] c. Parentheses () d. Quotation marks ""
-
Analyze the air pollution and mortality data in Table B. 15 using all possible regressions. Evaluate the subset models using the \(R_{p}^{2}, C_{p}\), and \(M S_{\text {Res }}\) criteria. Justify...
-
The 2009 financial statements of 3M Company report net sales of $23.1 billion. Accounts receivable (net) are $3.2 billion at the beginning of the year and $3.25 billion at the end of the year....
-
The CDS curve in question is shown: Table 1: CDS Curve CDS Maturity (Years) 1234567 Quoted Flat CDS Spread (bp) 30 40 50 60 70 80 90 8 100 9 110 10 120 The CDS spreads shown in this table are Quoted...
-
A solid plastic sphere falls towards the Earth. The diagram below shows the speed-time graph of the fall up to the point where the sphere hits the Earth's surface. a. Describe in detail the motion of...
-
Chloroform, CHCl 3 , a volatile liquid, was once used as an anesthetic but has been replaced by safer compounds. Chloroform boils at 61.7C and has a heat of vaporization of 31.4 kJ/mol. What is its...
-
An element crystallizes with a simple cubic lattice with atoms at all the lattice points. If the radius of the atom is 200. pm, what is the volume of the unit cell? a. 8.00 10 6 pm 3 b. 6.40 10 7...
-
Why should we be interested in the magnitude of the thrust force in grinding? Explain.
-
Who issues warrants? Multiple choice question. Investment bankers Stock exchanges The firm Stock brokers
-
x+5 x-2 Subtract and simplify: x-3 x+7
-
A company's free cash flow is $6,500. The average amount of debt maturing over the next five years is $5,405. What is the cash flow adequacy ratio? a. 20.3% b. 83.2% c. 120.3% d. 16.8%
-
If the net income from an investment is expected to be 75,000; 100,000; 125,000; and 120,000 for the next four year and the investment cost $600,000 what is the average accounting rate of return...
-
K Solve the following equation. 7 1 X- 8 x= (Type an integer or a simplified fraction.)
-
Distinguish between the following. a. The gross estate and the taxable estate. b. The taxable estate and the tax base. c. The gross estate and the probate estate.
-
On October 31 Juanita Ortega, owner of Outback Guide Service, received a bank statement dated October 30. Juanita found the following: 1. The checkbook has a balance of $2,551.34. 2. The bank...
-
What are the differences between a bonding and an antibonding molecular orbital of a diatomic molecule?
-
Describe the formation of molecular orbitals resulting from the interaction of two 2p orbitals.
-
Describe the bonding in O3, using molecular orbital theory. Compare this with the resonance description.
-
Why are gains/losses added back to (or deducted from) the cash from operations section of a cash flow statement?
-
Combination A, B, C, and F is the fifth most valuable with an NPV of $169,000. how did you came up with it?
-
Using a suitable example explain how you would check that equipment is ready for shut down to commence?

Study smarter with the SolutionInn App


