Which of the following two compounds has the shorter carbonoxygen bond? H :0: - - H Methanol
Question:
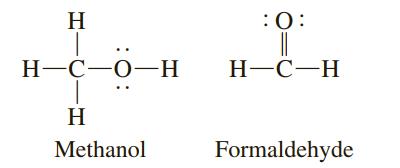
Which of the following two compounds has the shorter carbon–oxygen bond?
Transcribed Image Text:
H :0: Н-С—О—Н Н-С—Н H Methanol Formaldehyde
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
Formaldehyde has a shorter ca...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Sciences questions
-
which of the following in biological compounds are sufficiently electronegative to serve as strong donors in a hydrogen bond hydrogen and oxygen oxygen and nitrogen nitrogen and hydrogen hydrogen and...
-
The following two compounds each exhibit two heteroatoms (one nitrogen atom and one oxygen atom). In compound A, the lone pair on the nitrogen atom is more likely to function as a base. However, in...
-
Which of the following two compounds would eliminate HBr more rapidly in basic solutions? CH2Br or CH2Br
-
In Exercises 1318, find the average rate of change of the function from x 1 to x 2 . f(x)=x from x = 4 to x = 9
-
Explain how the implied repo rate on a spread transactions differs from that on a nearby futures contracts?
-
What type of file does Scene Builder save a GUI to?
-
A manufacturer of smart phones has the following probability distribution for the number of defects per phone: (a) Determine the probability of 2 or more defects. (b) Is a randomly selected phone...
-
Lessack Auctioneers advertised an auction sale that was open to the public and was to be conducted with reserve. Gordon attended the auction and bid $100 for a work of art that was worth much more....
-
Requirement 1. Determine the amount that would be reported in ending merchandise inventory on October 15 using the FIFO inventory costing method. Enter the transactions in chronological order,...
-
A digital audio system is designed to minimize the effect of disturbances as shown in Figure E4.2. As an approximation, we may represent G(s) = K2. (a) Calculate the sensitivity of the system due to...
-
Give resonance descriptions for the following: a. C 2 O 4 2- b. CH 3 NO 2
-
One of the following compounds has a carbonnitrogen bond length of 116 pm; the other has a carbonnitrogen bond length of 147 pm. Match a bond length with each compound. H H -N H-C-C=N: H H H...
-
Solve the given inequalities. Graph each solution. 0 3 2x 6
-
Megraf is a large manufacturing organisation which is structured with a numberof supervisors each responsible for their own work production unit. Supervisors report to a team of senior managers who...
-
Express your opinion regarding how technology advances (e.g.,robotic processing automation, blockchain, artificial intelligence, data analytics,) will impact the auditing field. Discuss which...
-
Assignment Summary: In this assignment, you are assisting a reporter who is writing a story to raise awareness around the 215 children found in a mass grave at Kamloops Indian Residential School....
-
Clara is a 14-year-old student presenting significant academic delays as compared to her peer group. She has just arrived in our community and has just been enrolled in school with no accompanying...
-
into the profession of nursing is a long and significant process that takes place over years of formal and information training. OA. Professionalization B. Socialization C. Dominance D. Negotiation
-
In 2013, Gray Corporation, a calendar year C corporation, has a $75,000 charitable contribution carryover from a gift made in 2008. Gray is contemplating a gift of land to a qualified charity in...
-
The following processes constitute the air-standard Diesel cycle: 12: isentropic compression,23: constant-volume energy addition (T and P increase),34: constant-pressure energy addition (v...
-
Commercially, sulfuric acid is obtained as a 98% solution. If this solution is 18 M, what is its density? What is its molality?
-
Phosphoric acid is usually obtained as an 85% phosphoric acid solution. If it is 15 M, what is the density of this solution? What is its molality?
-
A compound of manganese, carbon, and oxygen contains 28.17% Mn and 30.80% C. When 0.125 g of this compound is dissolved in 5.38 g of cyclohexane, the solution freezes at 5.28C. What is the molecular...
-
How does the central bank imply Bank Rate fluctuations as the monetary policy mechanism? Explain.
-
1. Calculate the NPV and Profitability Index for each of the following projects, assuming the cost of capital is 7%. YEAR iPad Pro iPhone 9 I 0 -36,000 -11,500 1 18,000 0 2 12,600 7,500 3 4,500 4,000...
-
State the dividend irrelevance proposition. What are the assumptions behind this proposition? Explain why this proposition does not hold in the real world. (20 marks)

Study smarter with the SolutionInn App


