(a) Will a plot of R(r) for the 1s atomic orbital of He + be identical to...
Question:
(a) Will a plot of R(r) for the 1s atomic orbital of He+ be identical to that of the H atom (Fig. 1.5a)?
(b) On the same axis set, sketch approximate representations of the function 4πr2R(r)2 for H and He+.
Figure 1.5a.
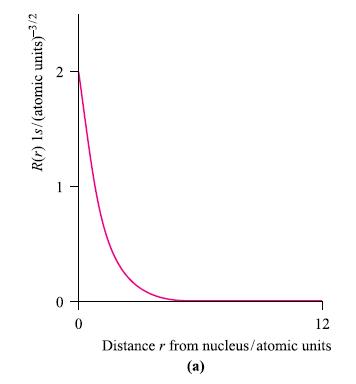
Transcribed Image Text:
R(r) 1s/(atomic units)-3/2 2 1 0 0 12 Distance from nucleus/atomic units (a)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a No a plot of Rr for the 1s atomic orbital of He will not be identical to ...View the full answer

Answered By

Qurat Ul Ain
Successful writing is about matching great style with top content. As an experienced freelance writer specialising in article writing and ghostwriting, I can provide you with that perfect combination, adapted to suit your needs.
I have written articles on subjects including history, management, and finance. Much of my work is ghost-writing, so I am used to adapting to someone else's preferred style and tone. I have post-graduate qualifications in history, teaching, and social science, as well as a management diploma, and so am well equipped to research and write in these areas.
4.80+
265+ Reviews
421+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A student needs to measure the drag on a prototype of characteristic length dp moving at velocity Up in air at sea-level conditions. He constructs a model of characteristic length dm, such that the...
-
4. Let g(r) be a continuous function on (0, oo). Solve the IVP: ry"- ry-g(1), IE (0, 00), y(1)-e,y(1)-0.
-
A consumer electronics company is comparing the brightness of two different types of picture tubes for use in its television sets. Tube type A has mean brightness of 100 and standard deviation of 16,...
-
an unadjusted trial balance as of December 31, 2020. (6points)Account TitleBalanceDebitCreditCash36,910Accounts Receivable5,750Office Supplies1,400 PART1: The following transactions occurred for...
-
Mel Hollingsworth recently left his job at a local pool company to open his own pool and spa maintenance business. Mel Hollingsworth took all of the money he and his wife had in their personal...
-
Calculate Gillian Pool and Spa Supplies average annual compound growth rate of sales and analyze its earnings performance for the past 5 years.
-
Based on the following pedigree for a trait determined by a single gene (affected individuals are shown as filled symbols), state whether it would be possible for the trait to be inherited in each of...
-
The completed worksheet for Vasquez Corporation as of December 31, 2016, after the company had completed the first month of operation, appears across the tops of pages 146147. INSTRUCTIONS 1. Prepare...
-
3. You have been provided with the information listed below for the City of XYZ. 2024 2025 market value of taxable property $1.3 billion increases by 2.0% from 2024 exemptions $26 million increases...
-
How is the (a) Energy (b) Size of an ns atomic orbital affected by an increase in n?
-
Using the Bohr model, determine the values of the radii of the second and third orbits of the hydrogen atom.
-
A triangular plate of narrow rectangular cross-section and uniform thickness carries a uniformly varying loading along its top edge as shown. Verify that the Airy stress function: p=[a14 cos+b14 sin...
-
Calculating Annuities You are planning to save for retirement over the next 30 years. To do this, you will invest 500 a month in a share account and 500 a month in a bond account. The return of the...
-
Future Value What is the future value in 4 years of 1,000 invested in an account with a stated annual interest rate of 10 per cent, (a) Compounded annually (b) Compounded semi-annually (c) Compounded...
-
When a new, faster computer chip is introduced, demand for computers using the older, slower chips decreases. Simultaneously, computer makers increase their production of computers containing the old...
-
The following graph shows inflation and unemployment rates for Canada for the period between 1970 and 2012. Does this graph show evidence in favor of the Phillips curve? 14 12 10 8 6 4 1 O 2 Canada...
-
Assuming that Okuns law is given by U U n = 0.75 (Y Y P ) and that the Phillips curve is given by = e - 0.6 (U U n ) + , a) Obtain the short-run aggregate supply curve if expectations are...
-
Explain why warranty expenses and the actual costs incurred with respect to warranties often do not occur in the same period.
-
Define deferred revenue. Why is it a liability?
-
The hydroxoacid Si(OH) 4 is weaker than H 2 CO 3 . Write balanced equations to show how dissolving a solid M 2 SiO 4 can lead to a reduction in the pressure of CO 2 over an aqueous solution. Explain...
-
In the gas phase, the base strength of amines increases regularly along the series NH 3 < CH 3 NH 2 < (CH 3 ) 2 NH < (CH 3 ) 3 N. Consider the role of steric effects and the electron-donating ability...
-
Assign oxidation numbers for each of the elements participating in the following reactions. 2NO(g) + O(g) 2NO(g) 2 Mn+ (aq) + 2HO(l) MnO (s) + Mn+ (aq) + 4H*(aq) LiCoO (s) + C(s) LiC (s) + CoO (s)...
-
5:07 19 Ci A rocket is launched at an angle of 59.0 above the horizontal with an initial speed of 102 m/s. The rocket moves for 3.00 s along its initial line of motion with an acceleration of 32.0...
-
9. An object (A) of mass m = 18.0 kg is moving due east with a speed V = 8.00 m/s, while object (B) of mass mg=29.0 kg is moving in a direction that makes angle of 60 north of west with a speed VB =...
-
A thin rod of length L and mass M is pivoted about its center as shown below. A small point- like mass (of mass m) is attached to the left end of the rod. A small point-like mass (of mass 2m) is...

Study smarter with the SolutionInn App


