Calculate the equilibrium constant of the reaction from the standard potentials Aut(aq) + 2 CN- (aq) [Au(CN)](aq)
Question:
Calculate the equilibrium constant of the reaction
![]()
from the standard potentials
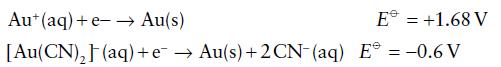
Transcribed Image Text:
Aut(aq) + 2 CN- (aq) [Au(CN)₂](aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
To calculate the equilibrium constant Keq for the given reaction we can use the Nernst equation The Nernst equation relates the standard electrode pot...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
Use electrode potentials to calculate the equilibrium constant at 25oC for the reaction See Appendix I for data. cio4(aq) + cio,-(aq)-2010,-(aq)
-
From the following reduction potentials (a) Calculate the equilibrium constant for .I2 (aq) + I- I-3 (b) Calculate the equilibrium constant for I2 (aq) + I- I-3 (c) Calculate the solubility (g/L) of...
-
Leno Company used regression analysis to predict the annual cost of indirect materials. The results were as follows: Indirect Materials Cost Explained by Units Produced Constant 14,885 Standard error...
-
A voltage V is applied to a dc electric motor. The armature winding resistance is equal to R. At what value of current flowing through the winding will the useful power of the motor be the highest?...
-
Multiple-Choice Questions 1. Microsoft found that instead of producing a DVD player and a gaming system separately, it is cheaper to incorporate DVD playing capabilities in their new version of the...
-
A tablet has 4 choices for an operating system, 3 choices for a screen size, 4 choices for a processor, 6 choices for memory size, and 3 choices for a battery. How many ways can you customize the...
-
What would happen to the SML graph in Figure 8.8 if expected inflation increased or decreased? Figure 8.8 268 269 270 271 272 273 274 275 A Required Rate of Return TH-13.0% SML: r, RF+RPM * b D E F H...
-
Nona Inc., which produces a single product, has prepared the following standard cost sheet for one unit of the product. Direct materials (8 pounds at $2.50 per pound) ....$20 Direct labor (3 hours at...
-
4. The effects of the German reunification in the Solow world. Imagine that West Germany in 1989 was well represented by the following differential equation. k =ska (8+n+g)k Let's assume that at that...
-
From the following Latimer diagram (which does not correspond to standard conditions), calculate the value of E for the reaction 2HO 2 (aq) O 2 (g) + H 2 O 2 (aq). Comment on the thermodynamic...
-
In their article Enzymes and bio-inspired electrocatalysts in solar fuel devices (Energy Environ. Sci., 2012, 5, 7470), Woolerton et al. use a generic Frost diagram (Fig. 6.21) to illustrate the...
-
Employee monitoring often collects information on work-related activities, but it can also collect an employees personal information not linked to their work. How would you manage employees who want...
-
Question Content AreaIf merchandise that cost the company $17,000 is expected to be returned in the coming year, the required adjustment _____. a. increases Estimated Returns Inventory and decreases...
-
The goal is to reduce wastefulness and develop a tight, efficient budget. The management team knows that this will take time, so they plan to allow more time and additional resources in the budget...
-
Cormac purchased furniture from a local business with no payments for 12 months at 0% interest. Six months later, he was told he owed the original amount, plus interest and late fees. When he finally...
-
Plot the number 0.0015 on the log scale below. 105 104 103 102 101 100 101 102 103 104 105
-
Question 46 (15 points) Complete the following java code as stated below: import java.util.Scanner; public class Example { //----- //This program reads the amount of money spent by customers and...
-
Calculate E cell for the reaction CH 3 OH(l) + 3/2O 2 (g) CO 2 (g) + 2H 2 O(l) Using values of G o f in Appendix 4. Will E cell increase or decrease with an increase in temperature?
-
Use Stokes' Theorem to evaluate f(y+sin x) dx+(z+cos y) dy+rdz, where C is the rve r(t) = (sint, cost, sin 2t), t = [0, 2].
-
Suggest possible solid state precursors for the formation of the following compounds by pyrolysis reactions: (a) BiCaVO 5 ; (b) The Mo(VI) oxide CuMo 2 YO 8 ; (c) Li 3 InO 3 ; (d) Ru 2 Y 2 O 7 .
-
(a) Describe the relationship between the structures of MgB 2 and graphite. (b) How does the electronic structure of MgB 2 differ from that of graphite, and how does this affect the properties of the...
-
(a) Describe the layered structure of FeSe. (b) How are the structures of NaFeAs and LaOFeAs related to that of FeSe? (c) Whereas LaOFeAs is not superconducting, LaO 1x F x FeAs is a superconductor...
-
Thornton Manufacturing Company was started on January 1 , 2 0 1 8 , when it acquired $ 8 1 , 0 0 0 cash by issuing common stock. Thornton immediately purchased office furniture and manufacturing...
-
1. Design a TM to perform circular left-shift operation. 2. Design a TM to accept the language (0a, 1ab, 2bab 1}. Draw the state-transition diagram, and explain how your TM operates. 3. Let w =...
-
Sachs Brands's defined benefit pension plan specifies annual retirement benefits equal to 1 . 6 % times service years times final year's salary, payable at the end of each year. Angela Davenport was...

Study smarter with the SolutionInn App


