Calculate the overall formation constant for [Fe(CN) 6 ] 3 , given that the overall formation constant
Question:
Calculate the overall formation constant for [Fe(CN)6]3−, given that the overall formation constant for [Fe(CN)6]4− is ≈ 1032, and that:
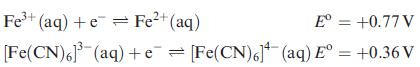
Transcribed Image Text:
Fe³+ (aq) + e Fe²+ (aq) [Fe(CN)6]³(aq) E = +0.77 V + e[Fe(CN)6] (aq) E° = +0.36 V
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
To calculate the overall formation constant for FeCN63 we can use the Nernst equation which relates ...View the full answer

Answered By

Aketch Cindy Sunday
I am a certified tutor with over two years of experience tutoring . I have a passion for helping students learn and grow, and I firmly believe that every student has the potential to be successful. I have a wide range of experience working with students of all ages and abilities, and I am confident that I can help students succeed in school.
I have experience working with students who have a wide range of abilities. I have also worked with gifted and talented students, and I am familiar with a variety of enrichment and acceleration strategies.
I am a patient and supportive tutor who is dedicated to helping my students reach their full potential. Thank you for your time and consideration.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The firm gets 70% of its capital from common stock and 30% from debt. The debtholders required rate of return is 8%. The equity holders required rate of return is 13% and the firms tax rate is 20%....
-
The overall formation constant for HgI42- is 1.0 Ã 1030. That is, [Hgl,-) [Hg 1.0 x 1030 =
-
Use the solubility-product constant for Cr(OH) 3 (K sp = 6.7 10 -31 ) and the formation constant for Cr(OH) 4 from Table 17.1 to determine the concentration of Cr(OH) 4 in a solution that is...
-
Which of the following options are available for creating a policy in Qualys Policy Compliance? (Choose three) A, Create from Host B, Create from Scratch C, Import from Library D, Import from CSV File
-
Global competition is determined in part by both efficiency and innovation. To develop a foreign market entry strategy for your company, a colleague informed you of a competitiveness report that is...
-
Access the GASB website (www.gasb.org). List and summarize the GASB Concept Statements.
-
Dr. Walter Sullivan was one of several plastic surgeons in Las Vegas visited by Julie Jones. Jones, an exotic dancer, sought plastic surgery to improve her ability to make money in her profession....
-
Kishkumen Company had the following capital structure at December 31, 2011 and 2012: The following additional information is available. (a) The conversion terms of the preferred stock and bonds at...
-
Electronex Corporation currently employs four workers. Tom is capable of producing 20 transistor radios or 5 cellular telephones.Sue can produce 8 radios or 8 phones.Jane can produce 4 radios or 16...
-
Using data from Table 8.1, write down the spontaneous cell process, and calculate E o cell and G o for the following combinations of half-cells: Data from Table 8.1 (a) Ag+ (aq) +e=Ag(s) (b) Br (aq)...
-
Using appropriate data from eqs. 8.43 to 8.47, confirm the value of E o given for eq. 8.48. Equations 12+ (aq) + 2e Mn(s) Mn+ [MnO4] (aq) + e[MnO4) E = -1.19 V (aq) E = +0.56 V 2+ MnO (s) + 4H+ (aq)...
-
What are a firms two principal sources of financing? Of what do these sources consist?
-
Imagine that you were a new CFO of Beily Inc., a childrens bicycle manufacturer. The president, Mr. Zhao, started the business 2 years ago. The firm manufactures two types of products, bicycles for...
-
Top Performance has been operating for several years in the outskirts of Ostersund, Sweden, and is a new manufacturer of a top-of-the-line outdoor jacket. You are starting an internship as assistant...
-
The debt and equity section of the TOTAL Group balance sheet is shown here (in million euros). The current market price of the common shares is 200. Reconstruct the financial statement assuming that...
-
The General Meeting of Remy Cointreau SA, listed on Euronext Paris, held in July 2015, set the overall dividend at 1.53 per share. The Meeting decided to grant every shareholder payment of the...
-
Door Company sells products in cash and in credit as follows: cash sales are 60 percent of total sales, and credit sales are 40 percent. 10 percent of the credit sales are collected in the month of...
-
Saputo Inc. is a Montreal-based producer and distributor of dairy products, including cheese and yogurt products. According to the companys website, it is one of the top ten dairy processors in the...
-
Express mass density in kg/m3 and weight density in lb/ft3. 1. Find the mass density of a chunk of rock of mass 215 g that displaces a volume of 75.0 cm3 of water. 2. A block of wood is 55.9 in. x...
-
In their review on new commercial applications of Ln chemistry (Acc. Chem. Res., 2016, 49, 844), Guillou and coworkers explain how the coordination of different Ln(III) ions within metalorganic...
-
Predict what species are formed when Pu metal is dissolved in dilute HCl and the nature of the solid product that forms when HF is subsequently added.
-
Explain why UF 3 and UF 4 are high melting point solids whereas UF 6 sublimes at 57C.
-
1. when trying to determine a strategy for factoring trinomials of the form ax + bx + c,a 1, the binomial factors (mx+p) and (nx + q) were multiplied to give mnx+(mq+np)x + pq. Match each expression...
-
In this question, do not make any assumptions on the price process of the underlying. (a) [15 marks] Let C(K) be the price of a European call option with strike price K. Consider strike prices K1, K2...
-
Round all of your answers to the nearest cent when appropriate to do so. Some questions are programmed to allow for slight variations in the answers due to rounding errors, BUT it is important that...

Study smarter with the SolutionInn App


