In each of the following reactions, relate starting materials and products by the processes of reduction, oxidation,
Question:
In each of the following reactions, relate starting materials and products by the processes of reduction, oxidation, disproportionation or no redox change. In some reactions, more than one process is taking place.
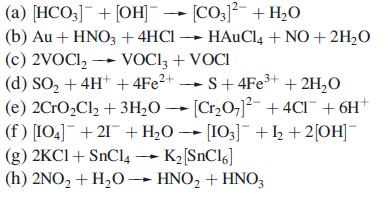
Transcribed Image Text:
(a) [HCO3] + [OH] → [CO3)² + H₂O (b) Au + HNO3 + 4HC1 →→ HAuCl4 + NO + 2H₂O (c) 2VOCI₂ VOCI3 + VOCI (d) SO₂ + 4H+ + 4Fe²+ (e) 2CrO₂Cl₂ + 3H₂O (f) [104]+21+ H₂O → [103]¯¯ +1₂ + 2[OH]¯ (g) 2KCI+ SnCl4 → K₂ [SnC16] (h) 2NO₂ + H₂O → HNO₂ + HNO3 S +4Fe³+ + 2H₂O [Cr₂O7]2+4Cl¯ + 6H+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
a HCO3OH CO32HO This reaction involves reduction and oxidation Carbon in HCO3 is oxidized wh...View the full answer

Answered By

Chandrasekhar Karri
I have tutored students in accounting at the high school and college levels. I have developed strong teaching methods, which allow me to effectively explain complex accounting concepts to students. Additionally, I am committed to helping students reach their academic goals and providing them with the necessary tools to succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Case Study: Quick Fix Dental Practice Technology requirements Application must be built using Visual Studio 2019 or Visual Studio 2017, professional or enterprise. The community edition is not...
-
Each of the following scenarios requires the use of accounting information to carry out one or more of the following managerial activities: (1) Planning, (2) Control and evaluation, (3) Continuous...
-
Were Napoleon's territorial ambitions significantly different from pre-1799 conquests? If so, where?What were Napoleon's most significant domestic accomplishments in France? Consider the interesting...
-
After watching the video on GM in Shanghai, discuss the following: 1. How would you characterize the alliance between GM and SAIC Shanghai? 2. What has driven the GM alliance in Shanghai? 3. How is...
-
Betty Sims has $30,000 of adjusted gross income and $5,000 of medical expenses. She will be itemizing her tax deductions this year. The most recent tax year has a medical expenses floor of 10...
-
Fun-Tastic Shows, Inc., is a company that hosts carnivals and similar events. Susan Swartwood, Crystal Groth, and a minor (named in the case as M.G.S.) attended Fun-Tastics Rhododendron Festival in...
-
The Vang Hotel opened for business on May 1, 2012. Here is its trial balance before adjustment on May 31. Other data:1. Insurance expires at the rate of $450 per month.2. A count of supplies shows...
-
asap please 1. Determine a maintenance priority by calculating Preventive Maintenance Factors (PMFs) for the three pieces of equipment listed in the table below. Clearly state which piece of...
-
The commercial purification of copper metal is carried out in electrolytic cells. The anode is composed of impure (blister) copper, and the electrolyte is a mixture of aqueous CuSO 4 and H 2 SO 4 ....
-
Zinc/silver oxide button batteries are used in calculators and watches. Although the silver component makes them expensive, this is outweighed by their high performance. The battery uses KOH as the...
-
Pena Corporation incurred the following costs while manufacturing its product. Instructions (a) Identify each of the above costs as direct materials, direct labor, manufacturing overhead, or period...
-
AllCity, Inc., is financed 35% with debt, 6% with preferred stock, and 59% with common stock. Its pretax cost of debt is 6.5%, its preferred stock pays an annual dividend of $2.51 and is priced at...
-
Your firm has an ROE of 12.3%, a payout ratio of 24%, $638,700 of stockholders equity, and $415,000 of debt. If you grow at your sustainable growth rate this year, how much additional debt will you...
-
Mackenzie Company has a price of $32 and will issue a dividend of $2 next year. It has a beta of 1.5, the risk-free rate is 5.6%, and it estimates the market risk premium to be 4.8%. a. Estimate the...
-
Your firm currently has $92 million in debt outstanding with a 7% interest rate. The terms of the loan require the firm to repay $23 million of the balance each year. Suppose that the marginal...
-
Suppose the corporate tax rate is 30%. Consider a firm that earns $1000 before interest and taxes each year with no risk. The firms capital expenditures equal its depreciation expenses each year, and...
-
What are the two key ratios used to analyze inventory? Explain what each of the ratios tells us about a companys inventory.
-
The senior management at Davis Watercraft would like to determine if it is possible to improve firm profitability by changing their existing product mix. Currently, the product mix is determined by...
-
(a) Describe the two classes of inorganicorganic nanocomposites based on their bonding types. (b) Give one example of a nanocomposite in each class.
-
Discuss shape selectivity with respect to catalytic processes involving zeolites including mechanisms involving reactant, transition state, and product selectivity.
-
(a) What is the relevance of self-assembly to the fabrication of nanomaterials? (b) What role will it play in nanotechnology?
-
Solve the system. State whether the system is inconsistent or has infinitely many solutions. If the system has infinitely many solutions, write the solution set with y arbitrary. 2x+3y=3 4x+6y=6...
-
Find the derivatives of the following functions a) to c). Show your steps. a) y = f(x) = ex In (3x + 1) (5 points) b) y = f(x) = (5x+9)21 x3+4x+5 (5 points)
-
The interest rate on an auto loans has increased 50% over the past decade and now stands at 12%. What can you conclude?

Study smarter with the SolutionInn App


