In each redox reaction in problem 8.3, confirm that the net increases and decreases in oxidation states
Question:
In each redox reaction in problem 8.3, confirm that the net increases and decreases in oxidation states balance each other.
Data from Problem 8.3
Which of the following reactions are redox reactions? In those that are, identify the oxidation and reduction processes.
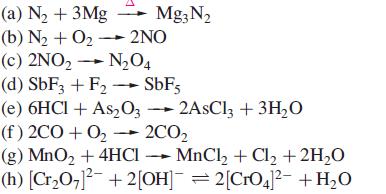
Transcribed Image Text:
(a) N₂ + 3Mg Mg3N₂ (b) N₂ + O₂- 2NO (c) 2NO₂ → N₂04 (d) SbF3 + F₂ → SbF5 (e) 6HCl + As2O32AsCl3 + 3H₂O (f) 2CO+O₂ → 2CO₂ (g) MnO₂ + 4HCI MnCl₂ + Cl₂ + 2H₂O (h) [Cr₂O7]²¯ + 2[OH]¯ = 2[CrO4]²¯ +H₂O
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (6 reviews)
To determine whether each reaction is a redox reaction and confirm that the net increases and decreases in oxidation states balance each other we need to identify the oxidation and reduction processes ...View the full answer

Answered By

Akash M Rathod
I have been utilized by educators and students alike to provide individualized assistance with everything from grammar and vocabulary to complex problem-solving in various academic subjects. I can provide explanations, examples, and practice exercises tailored to each student's individual needs, helping them to grasp difficult concepts and improve their skills.
My tutoring sessions are interactive and engaging, utilizing a variety of tools and resources to keep learners motivated and focused. Whether a student needs help with homework, test preparation, or simply wants to improve their skills in a particular subject area, I am equipped to provide the support and guidance they need to succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The commercial production of nitric acid involves the following chemical reactions: (a) Which of these reactions are redox reactions? (b) In each redox reaction identify the element undergoing...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Read the case study "Southwest Airlines," found in Part 2 of your textbook. Review the "Guide to Case Analysis" found on pp. CA1 - CA11 of your textbook. (This guide follows the last case in the...
-
Montage Pty Limited (Montage) is a resident private company and is not a base rate entity. Which of the following transactions would result in a debit entry to Montage's franking account? Payment of...
-
Using Peng Atlas Map 3.1, in your option would it be easier or more difficult to engage in competitive actions in those countries that are at the bottom?
-
Rachael is verifying the accuracy and amount of information contained in the employee records for her company. Which of the following items should be present in the employee information? 1. Job title...
-
Sabrina Ederly worked as a reporter and contributing editor for Rolling Stone magazine. Ederly intended to write an article for the magazine that focused on the alleged rape of a University of...
-
Consider the following network for a small maintenance project (all times are in days; network proceeds from node 1 to node 7): a. Draw an arrow diagram representing the project. b. What is the...
-
Assume a simultaneous open market purchase of 100 million from the Bank of England and a repayment of a discount loan of 5 million from Bank A to the Bank of England. Show the overall change in their...
-
(a) Calculate E Ag + /Ag for a half-cell in which the concentration of silver(I) ions is 0.1moldm 3 (T = 298 K). (b) Are silver(I) ions more or less easily reduced by zinc in this solution than under...
-
Consider the half-reaction: If the ratio of concentrations of [MnO 4 ] : Mn 2+ is 100:1, determine E at pH values of (a) 0.5; (b) 2.0; (c) 3.5 (T = 298 K). Over this pH range, how does the ability...
-
Assume you are in the 35 percent tax bracket and purchase a 2.8 percent, tax-exempt municipal bond. Use the formula presented in this chapter to calculate the taxable equivalent yield for this...
-
The Ohio Valley Steel Corporation has borrowed $5.1 million for one month at a stated annual rate of 8.7%, using inventory stored in a field warehouse as collateral. The warehouser charges a $5100...
-
Hardmon Enterprises is currently an all-equity firm with an expected return of 11.2%. It is considering borrowing money to buy back some of its existing shares, thus increasing its leverage. a....
-
Arnell Industries has $15 million in permanent debt outstanding. The firm will pay interest only on this debt. Arnells marginal tax rate is expected to be 35% for the foreseeable future. a. Suppose...
-
Rally, Inc., is an all-equity firm with assets worth $56 billion and 7 billion shares outstanding. Rally plans to borrow $22 billion and use these funds to repurchase shares. The firms corporate tax...
-
You happen to be checking the newspaper and notice an arbitrage opportunity. The current stock price of Intrawest is $22 per share and the one-year risk-free interest rate is 7%. A one-year put on...
-
Sanjay Supplies Limited is concerned about its ability to pay its debts. Selected financial information follows. Required: Analyze the information and explain why Sanjay is experiencing problems with...
-
Periwinkle Company is a multinational organization. Its Parts Division is located in Lavender Land, while its Assembly Division is located in North Orchid. During the current year Periwinkle Companys...
-
Describe the inorganic chemistry involved in the operation of a lambda sensor (an exhaust-gas oxygen sensor) in a vehicle engine.
-
Identify the likely products of the reactions (a) Li,CO,+CoO_800C,0_ (b) 2 Sr(OH) +WO+MnO 900C,0,
-
Outline how you could prepare samples of (a) MgCr 2 O 4 , (b) LaFeO 3 , (c) Ta 3 N 5 , (d) LiMgH 3 , (e) KCuF 3 , (f) The zeolite A analogue with Ga replacing Al, Na 12 [Si 12 Ga 12 O 48 ]nH 2 O.
-
Perform the indicated operations. Express your answer in reduced form. 9 2 (x-5)(x + 1) x-5
-
rephrase: Secondly, financial resources are crucial to cover expenses such as instructional materials, technology tools, and any additional costs associated with implementing the module. Careful...
-
What events of the 1980s prompted public demand for governments to "get tough" on fraudulent businesses and negligent accountants? a. stock market crash b. bank failures in the U.S. c. layoffs of...

Study smarter with the SolutionInn App


