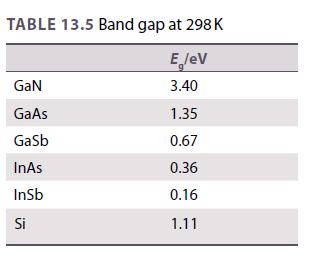
Use the data in Table 13.5 to estimate a band gap for the semiconductor GaP. Use this
Question:
Use the data in Table 13.5 to estimate a band gap for the semiconductor GaP. Use this to calculate the wavelength of light that is emitted when an electron drops from the conduction band to the valence band. Is this consistent with the observed green light of gallium phosphide LEDs?
Table 13.5.
Transcribed Image Text:
TABLE 13.5 Band gap at 298 K E lev 3.40 GaN GaAs Gasb InAs InSb Si 1.35 0.67 0.36 0.16 1.11
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The band gap energy Eg of a semiconductor can be related to the wavelength of light it emits or abso...View the full answer

Answered By

Sayee Sreenivas G B
I have extensive tutoring experience, having worked as a private tutor for over three years. I have tutored students from different academic levels, including high school, undergraduate, and graduate levels. My tutoring experience has taught me to be patient, attentive to student needs, and effective in communicating difficult concepts in simple terms.
I have a strong background in statistics, probability theory, data analysis, and data visualization. I am proficient in using statistical software such as R, Python, and SPSS, which are commonly used in academic research and data analysis. Additionally, I have excellent communication and interpersonal skills, which enable me to establish rapport with students, understand their learning styles, and adapt my teaching approach to meet their needs.
I am passionate about teaching and helping students achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
Light of wavelength 1.03 107 m is emitted when an electron in an excited level of a hydrogen atom undergoes a transition to the n = 1 level. What is the region of the spectrum of this light? What is...
-
Calculate the frequency (Hz) and wavelength (nm) of the emitted photon when an electron drops from the n = 4 to the n = 2 level in a hydrogen atom.
-
The maximum wavelength of light that a certain silicon photocell can detect is 1.11m. (a) What is the energy gap (in electron volts) between the valence and conduction bands for this photocell? (b)...
-
The net present value and internal rate of return desirability measures for two mutually exclusive investments being considered by Stockton Corporation to follow. Year NPV IRR R 161 14.60% S 138...
-
Panda Airlines Inc. has two divisions organized as profit centers, the Passenger Division and the Cargo Division. The following divisional income statements were prepared: The service department...
-
The CEO and CFO from Lawrence Hotels, Inc., are reviewing company performance for 2018. The income statement reports a 25% increase in net income over 2017. However, most of the increase resulted...
-
Redesign the fractionator of Example 6.8 for a reflux ratio that is twice the minimum. Determine the diameter of the tower, the height of packing in the stripping and rectifying sections, and the...
-
Given the following highway rerouting project, (a) Draw the network. (b) Find the ESs, LSs, and slacks. (c) Find the critical path. (d) If the project has a 1 1/2-year deadline for reopening, should...
-
In service partnerships, if properly booked for tax purposes pursuant to Treasury Regulations under IRC 731, partners who receive so-called "draw" allowances are not deemed to receive distributions...
-
Starting with B 10 H 14 and other reagents of your choice, give the equations for the synthesis of [Fe(nido-B 9 C 2 H 11 ) 2 ] 2 , and sketch the structure of this species.
-
Hydrolysis of 1 mol of a borohydride yields 15 mol of H 2 and 6 mol of B(OH) 3 . Identify the compound and suggest a structure.
-
Why is depreciation not represented as an event in the REA data model?
-
Analyze and interpret the complex accounting implications of a merger between two multinational corporations, each with unique accounting practices, tax structures, and financial reporting standards....
-
For a large boiler, you have the choice of the following energy resources. Calculate the point of use cost per GJ for each Natural gas at $ 3.00 per kg, efficiency 75% Electricity at $ 0.18/kWh,...
-
When preparing the bank reconciling, how are NSF treated? Explain.
-
Which type of codes are used to report services provided face-to-face by a physician or other qualified healthcare professional for the purpose of promoting health and preventing illness or injury.
-
If a large chilled water plant has 6 chillers and 5 of them are operating at 1 0 0 % , when the operator starts the 6 th chiller, to what percent do the first 5 chillers unload? What formula do you...
-
A RingCentral survey of 380 smartphone users showed that 152 of them said that their smartphone is the only thing they could not live without. Use a 0.01 significance level to test the claim that...
-
How does the organizational structure of an MNC influence its strategy implementation?
-
Comment on each of the following statements. (a) Ln 2+ complexes are strong reducing agents. (b) In the solid state, Cp 2 YbF(THF) exists as a bridged dimer, while Cp 2 YbCl(THF) and Cp 2 YbBr(THF)...
-
Give a short account of aspects of the organometallic compounds formed by the lanthanoids and actinoids and highlight major differences between families of organometallic complexes of the d- and...
-
Discuss the following: (a) Many actinoid oxides are non-stoichiometric, but few lanthanoid oxides are. (b) The ion [NpO 6 ] 5 can be made in aqueous solution only if the solution is strongly...
-
Question 3 [20 marks] a. [10 marks] You are given the following recurrence relation T(n) 27(n/2)+n for n>2, n=2*, 7(2)=2. Solve the above recurrence by the backward substitution method.
-
TASK NO. 1: REVIEW THE VOCABULARY FOR THE TYPOLOGIES Be sure to review the vocabulary for the typologies and make notes about what certain words mean. For example, if you don't know what the...
-
b. [10 marks] The pseudocode for the recursive binary search algorithm to search a key x in an array 4[1] containing n integers is shown below. RecursiveBinarySearch(4, x, L, r) if L>r then return -1...

Study smarter with the SolutionInn App


