Using data in Table 7.5, determine the relative solubilities of Ca(OH) 2 and Mg(OH) 2 and explain
Question:
Using data in Table 7.5, determine the relative solubilities of Ca(OH)2 and Mg(OH)2 and explain the relevance of your answer to the extraction of magnesium from seawater.
Table 7.5.
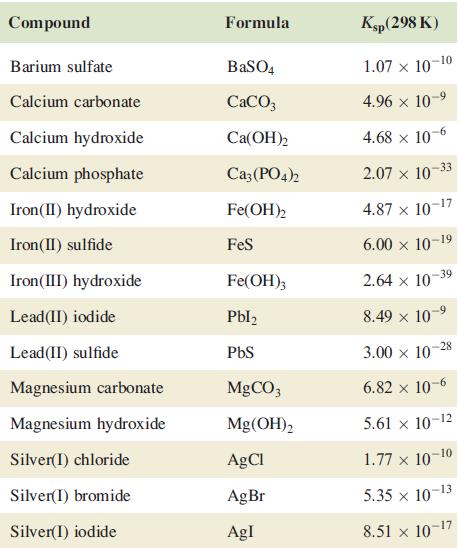

Transcribed Image Text:
Compound Barium sulfate Calcium carbonate Calcium hydroxide Calcium phosphate Iron (II) hydroxide Iron (II) sulfide Iron(III) hydroxide Lead (II) iodide Lead (II) sulfide Magnesium carbonate Magnesium hydroxide Silver(I) chloride Silver(1) bromide Silver(1) iodide Formula BaSO4 CaCO3 Ca(OH)2 Ca3(PO4)2 Fe(OH)2 FeS Fe(OH)3 Pbl₂ PbS MgCO3 Mg(OH)2 AgCl AgBr AgI Ksp (298 K) 1.07 x 10-10 4.96 × 10-⁹ 4.68 x 10-6 2.07 x 10 4.87 x 10-17 6.00 × 10-19 2.64 x 10-39 8.49 × 10-9 3.00 x 10 6.82 x 10-6 5.61 × 10-12 1.77 x 10-10 5.35 × 10-13 8.51 x 10-1 -17 -33 -28
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
Based on the data provided in Table 75 we can determine the relative solubilities of CaOH2 and MgOH2 by comparing their solubility product constants K...View the full answer

Answered By

Surendar Kumaradevan
I have worked with both teachers and students to offer specialized help with everything from grammar and vocabulary to challenging problem-solving in a range of academic disciplines. For each student's specific needs, I can offer explanations, examples, and practice tasks that will help them better understand complex ideas and develop their skills.
I employ a range of techniques and resources in my engaged, interesting tutoring sessions to keep students motivated and on task. I have the tools necessary to offer students the support and direction they require in order to achieve, whether they need assistance with their homework, test preparation, or simply want to hone their skills in a particular subject area.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Using data in Table 4.2 and assuming t/2R=0.07, compute the average circumferential stress in the ascending aorta, thoracic aorta, and femoral artery. Table 4.2: Values of the "pressure-strain", Ep,...
-
The accompanying stem and leaf plots were generated using MINITAB for the variables named "Popular Vote" and "Percent Vote." For Information: Refer to Exercises 1.51 and 1.52. Exercises 1.51 and 1.52...
-
The data listed in the following table relate to a study by Reiter and others (1981) concerning the effects of injecting triethyl-tin (TET) into rats once at age 5 days. The animals were injected...
-
When something burns, a. it combines with phlogiston b. it gives off phlogiston c. it combines with oxygen d. it gives off oxygen
-
Elite Classic Clothes is a retailer that sells to professional women in the northeast. The firm leases space for stores in upscale shopping centers, and the organizational structure consists of...
-
Find P(not B). Use the information that, for events A and B, we have P(A) = 0.8, P(B) = 0.4, and P(A and B) = 0.25.
-
True or False: If \(E R R>M A R R\), then \(I R R>E R R>M A R R\).
-
The December cash records of Duffy Insurance follow: Duffys Cash account shows a balance of $17,050 at December 31. On December 31, Duffy Insurance received the following bank statement: Additional...
-
Solve 1 17) log81 27 18) log 32 = 16 19) log40= 20) log101=
-
(a) Explain how face-sharing between M 6 O octahedra leads to compounds with stoichiometries of M 9 O 2 for M = Rb, and M 11 O 3 for M = Cs. (b) The suboxide Cs 7 O contains Cs 11 O 3 clusters....
-
Suggest explanations for the following observations. (a) Although Na 2 O 2 is described as being colourless, samples of Na 2 O 2 often appear to be very pale yellow. (b) NaO 2 is paramagnetic.
-
Define cell differentiation and morphogenesis, two processes that are critical for development.
-
Interview a faculty member or a businessperson who has worked abroad. Ask him or her to identify factors that facilitated or inhibited adaptation to the host environment. Ask whether more preparation...
-
Are there any conflicts that might arise between the issuer and the investment banker when setting the offering price on a stock issue?
-
Describe the net present social value model of capital budgeting.
-
Explain how the separation of ownership and control in organizations has led to an agency problem and agency costs.
-
For this exercise, research (individually or in small groups) a company with international operations and find out the kinds of entry strategies the firm has used. Present the information you find,...
-
You have just completed an interview with the newly formed audit committee of the Andrews Street Youth Centre (ASYC). This organization was created to keep neighborhood youth off the streets by...
-
Integration is a vital concept when applied in one?s life. Integrating your life means making ideal choices. Perfect choices on the other go in line with quality decisions. Quality decisions lead to...
-
The 31 P-MAS-NMR spectrum of solid PCl 5 shows two resonances, one of which has a chemical shift similar to that found for 31 P in the salt CsPCl 6 . Explain.
-
Explain why the 13 C-NMR spectrum of [Fe 3 (CO) 12 ] shows only a single peak at 212.5 ppm at room temperature while the IR spectrum shows the presence of both terminal and bridging carbonyl groups....
-
Explain the observation that the 19 F-NMR spectrum of the [XeF 5 ] anion consists of a central peak symmetrically flanked by two peaks, each of which is roughly one-sixth of the intensity of the...
-
Refer to the table below: Expected return, E(R) Standard deviation, o Correlation 3 Doors, Inc. 16% 31 Down Co. 9% 19 .40 Using the information provided on the two stocks in the table above, find the...
-
the accompanied three articles posted on some of these athlete's personal journeys and answer the following questions. How is it that some athletes that have been provided so much opportunity and in...
-
Elmdale Enterprises is deciding whether to expand its production facilities. Although long-term cash flows are difficult to estimate, management has projected the following cash flows for the first...

Study smarter with the SolutionInn App


