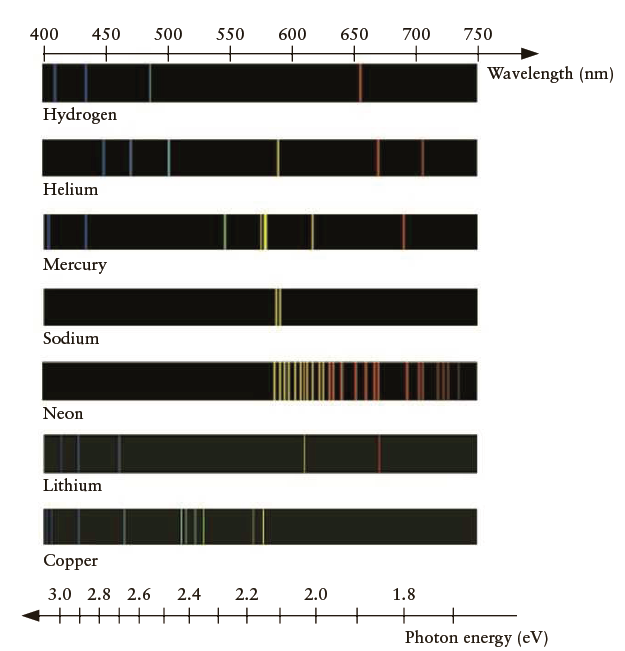
Compare the emission spectra of the elements hydrogen and helium (Figure 10.12). Which element emits photons of
Question:
Compare the emission spectra of the elements hydrogen and helium (Figure 10.12). Which element emits photons of red light that have the higher energy?
Figure 10.12
Transcribed Image Text:
650 750 400 450 500 550 600 700 Wavelength (nm) Hydrogen Helium Mercury Sodium Neon Lithium Copper 2.0 1.8 3.0 2.8 2.6 2.4 2.2 + + Photon energy (eV)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
The red line in the helium spectrum is closer to ...View the full answer

Answered By

Ajeet Singh
Hi there! Are you looking for a committed, reliable, and enthusiastic tutor? Well, teaching and learning are more of a second nature to me, having been raised by parents who are both teachers. I have done plenty of studying and lots of learning on many exciting and challenging topics. All these experiences have influenced my decision to take on the teaching role in various capacities. As a tutor, I am looking forward to getting to understand your needs and helping you achieve your academic goals. I'm highly flexible and contactable. I am available to work on short notice since I only prefer to work with very small and select groups of students. Areas of interest: Business, accounting, Project management, sociology, technology, computers, English, linguistics, media, philosophy, political science, statistics, data science, Excel, psychology, art, history, health education, gender studies, cultural studies, ethics, religion. I am also decent with math(s) & Programming. If you have a project you think I can take on, please feel welcome to invite me, and I'm going to check it out!
5.00+
4+ Reviews
24+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physics questions
-
Identifying Isotopes by Spectra Different isotopes of the same element emit light at slightly different wavelengths. A wavelength in the emission spectrum of a hydrogen atom is 656.45 nm; for...
-
Laser atomic fluorescence excitation and emission spectra of sodium in an air-acetylene flame are shown here. In the excitation spectrum, the laser (bandwidth 0.03 nm) was scanned through various...
-
Hydrogen sulfide is composed of two elements: hydrogen and sulfur. In an experiment, 6.500 g of hydrogen sulfide is fully decomposed into its elements. (a) If 0.384 g of hydrogen is obtained in this...
-
What are the explanations given for slow progress towards full acceptance of ISAs?
-
Discuss the reasons behind the new expansionism that characterized US foreign policy in 1890's.
-
Which of the following is designed primarily to improve the efficiency of financial reporting? a. XML b. XBRL c. IFRS d. The balanced scorecard
-
Construct a data set that has the given statistics. n = 5 x =12 s = 0
-
Maurice Browne recorded the following times assembling a watch. Determent (a) The average time (b) The normal time (c) The standard time taken by him, using a performance rating of 95% and a personal...
-
How does Process Analytical Technology (PAT) support real-time monitoring and control in bioprocessing, and what impact does it have on process optimization and product consistency ?
-
SUPPLY CHAIN MANAGEMENT W.W. Grainger and McMaster-Carr: MRO Suppliers W.W. Grainger and McMaster-Carr sell maintenance, repair, and operations (MRO) products. Both companies have catalogs and web...
-
Discuss what is meant by the term ionization. Give two ways by which an atom might acquire enough energy to become ionized.
-
If an astronomer examines the emission spectrum from luminous hydrogen gas that is moving away from Earth at a high speed and compares it to a spectrum of hydrogen seen in a laboratory on Earth, what...
-
Considering Section 7.1, suppose we began the analysis to find E = E 1 + E 2 with two cosine functions E 1 = E 01 cos (Ït + α 1 ) and E 2 = E 02 cos (Ït + α 2 )....
-
Learned optimism has been identified by psychologist Martin Seligman as a prerequisite for being successful personally and professionally. Access the Positive Psychology home page...
-
In terms of leadership strategy and communication styles, compare and contrast the communication and behavior styles of Jeff Bezos and Elon Musk. Identify the similarities and the differences in...
-
Using a negotiations worksheet, plan your responses to the following statements made by the purchasing manager: (a) We are concerned that you will not be able to meet our quality requirements; (b)...
-
Briefly describe the organization of this book.
-
Being a product and industry expert implies that the salesperson has knowledge of the most common product information categories. List the categories of product and industry knowledge a salesperson...
-
Why does the World View on page 449 say the undervalued yuan is more bane than boom?
-
(a) Given a mean free path = 0.4 nm and a mean speed vav = 1.17 105 m/s for the current flow in copper at a temperature of 300 K, calculate the classical value for the resistivity of copper. (b)...
-
Digesting fat produces 9.3 food calories per gram of fat, and typically 80% of this energy goes to heat when metabolized. (One food calorie is 1000 calories and therefore equals 4186 J.) The body...
-
An average sleeping person metabolizes at a rate of about 80 W by digesting food or burning fat. Typically, 20% of this energy goes into bodily functions, such as cell repair, pumping blood, and...
-
A certain heat engine operating on a Carnot cycle absorbs 150 J of heat per cycle at its hot reservoir at 135C and has a thermal efficiency of 22.0%. (a) How much work does this engine do per cycle?...
-
Hello, need help with making the program MealData.txt Avocado Egg Rolls 6.00 Parmesan-crusted Chicken 19.75 Strawberry Shortcake Pizookie 8.25 BBQ Tri-Tip Sliders 12.95 Spicy Peanut Chicken with Soba...
-
Solve the triangle shown to the right. Round the lengths of sides to the nearest tenth and angles to the nearest degree. 96 b=5 a=7 B A C C9.0 (Do not round until the final answer. Then round to the...
-
Assume you have been appointed to develop ethnic and multicultural marketing for a small chain of household appliance stores in large metropolitan area. There are several large concentrations of...

Study smarter with the SolutionInn App


