Consider an ethanol(1)/ethyl acetate(2) mixture with x 1 = 0.20, initially at 80C and 100 kPa. Describe
Question:
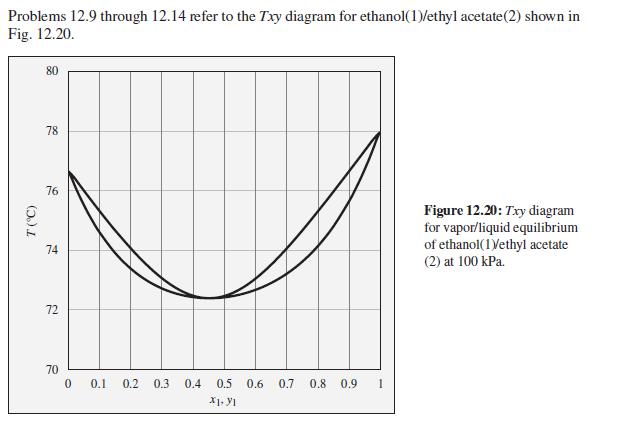
Consider an ethanol(1)/ethyl acetate(2) mixture with x1 = 0.20, initially at 80°C and 100 kPa. Describe the evolution of phases and phase compositions as the temperature is gradually reduced to 70°C.
Transcribed Image Text:
Problems 12.9 through 12.14 refer to the Txy diagram for ethanol(1)/ethyl acetate(2) shown in Fig. 12.20. 80 78 76 Figure 12.20: Txy diagram for vapor/liquid equilibrium of ethanol(1Vethyl acetate 74 (2) at 100 kPa. 72 70 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 1 X1. YI (5.)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
These are phase diagrams which show the composition of two ...View the full answer

Answered By

Hardik Dudhat
I am semi-qualified Chemical Engineering ,I have scored centum in accounting in my senior secondary and in my graduation. I have always helped my fellow students with their concerns on the subject, i have tutored on various tutoring sites in the past and also have taken home tuitions for degree and MBA students. As a tutor, I don't want my students to just get a solution, I want them to understand the concept and never have a doubt in that area thereon and i believe in excelling and not in educating.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
Describe the evolution of business-to-business (B2B) e-commerce.
-
Describe the evolution of ERP systems.
-
Describe the evolution of American culinary arts.
-
Will has a business loan of $100,000, it is for 7 years with an interest rate of 9%. How much interest would he pay over those 7 years?
-
Why does the interdependence of firms play a major role in oligopoly but not in perfect competition or monopolistic competition?
-
If interest rate parity is satisfied, there are no opportunities for covered interest arbitrage. What does this imply about the relationship between spot and forward exchange rates when the foreign...
-
Given the different needs of the HRP/workforce planning process discussed in this chapter, what types of data would you expect the HRIS data warehouse to contain?
-
Newfield Corp. holds a portfolio of trading securities. Suppose that on October 15, Newfield paid $85,000 for an investment in Turok shares to add to its portfolio. At December 31, the market value...
-
Open the jail.conf file and locate the default bantime and reset it to five minutes. Locate the number of retries in the ssh-iptables section and reset it to 3. Close and save the file. Restart the...
-
When a lipid bilayer is torn, why does it not seal itself by forming a hemi micelle cap at the edges, as shown in Figure Q101? Figure Q10-1 tear in bilayer seal with hemi-micelle cap
-
Consider an ethanol(1)/ethyl acetate(2) mixture with x 1 = 0.80, initially at 80C and 100 kPa. Describe the evolution of phases and phase compositions as the temperature is gradually reduced to 70C....
-
PVT data may be taken by the following procedure: A mass m of a substance of molar mass is introduced into a thermostated vessel of known total volume V t . The system is allowed to equilibrate, and...
-
Classify the quadrilateral. Explain your reasoning.
-
Consider the following project for Hand Clapper. The company is considering a 4-year project to manufacture clap-command garage door openers. This project requires an initial investment of 10 million...
-
You have just won a million-dollar lottery and will receive $50,000 at the end of each year for 20 years. What is the present value of the 20 annual payments to you today?
-
Explain what will happen to the present value of money one year from now if the market interest rate falls? What if the market interest rate rises?
-
Should welfare benefits be higher in Sweden and Denmark than in Romania? Defend your answer.
-
When a minimum wage is imposed, the number of people who lose their job will be less than the amount of any unemployment that may be created. Why is this?
-
In each part, find a single matrix that performs the indicated succession of operations: (a) Compresses by a factor of 1/2 in the x-direction, then expands by a factor of 5 in the y-direction (b)...
-
Find a polar equation for the curve represented by the given Cartesian equation. 4y 2 = x
-
Pressures up to 3.000fatmj are measured with a dead-weight gauge The piston diameter is 0.l7(in). What is the approximate mass in (lbm) of the weights required?
-
The reading on u mercury manometer at 70(oF) (open to the atmosphere at one end) is 25.62(in). The local acceleration of gravity is 32-243(ft)(s)-2. Atmospheric pressure is 29.86(in Hg). What is (he...
-
A nonconducting container tilled with 25 kg of water at 20 C is fitted with a stirrer, which is made to turn by gravity acting on a weight of mass 35 kg. The weight falls slowly through a distance of...
-
Provide a comprehensive analysis of the regulatory frameworks governing financial institutions, delineating the intricate interplay between prudential supervision, market conduct standards, and...
-
How does the forum meet organisational objectives? What vocabulary, tone, structure and style suits the audience? How will I: build trust develop positive working relationships ...
-
If a set of m+1 data points (x,y) are to be represent by a least squares fit of y=c logx derive an expression for c in terms of x and y.

Study smarter with the SolutionInn App


