Figure 3.3 suggests that the isochores (paths of constant volume) are approximately straight lines on a P-T
Question:
Figure 3.3 suggests that the isochores (paths of constant volume) are approximately straight lines on a P-T diagram. Show that the following models imply linear isochores.
(a) Constant-β, κ equation for liquids
(b) Ideal-gas equation
(c) Van der Waals equation
Figure 3.3
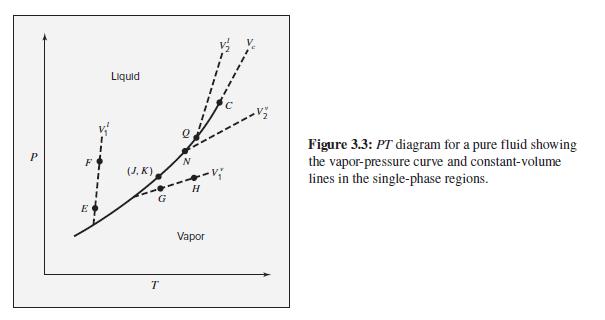
Transcribed Image Text:
Liquid Figure 3.3: PT diagram for a pure fluid showing the vapor-pressure curve and constant-volume lines in the single-phase regions. (J, K) G E Vapor T.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
Isochores constant volume lines are approximately straight lines on a PT diagram for some fluids We ...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
Van der Waals Equation and Critical Points (a) In p V- diagrams the slope p/V along an isotherm is never positive. Explain why. (b) Regions where p/V = 0 represent equilibrium between two phases;...
-
A certain gas obeys the van der Waals equation with a =0.76 m6 Pa mol-2, its volume is found to be 4.00 X 10-4 m3 mol-1 at 288 K and 4.0 MPa. From this information calculate the van der Waals...
-
Show that the van der Waals equation can be written as a cubic equation in the compressibility factor involving the reduced pressure and reduced temperature as 27 P 2 512T 3
-
Read each case carefully and, as determined in the Internal Revenue Code of Puerto Rico, identify the deductions, calculate the amount of deductions and what the determined contribution would be. and...
-
Why do you think companies invest resources in training their managers and employees in business ethics? Some people think that ethics should be learned within the family and by the time a person is...
-
Rush Corporation plans to acquire production equipment for $600,000 that will be depreciated for tax purposes as follows: year 1, $120,000; year 2, $210,000; and in each of years 3 through 5, $90,000...
-
Could an include use case have a primary actor? Explain.
-
At December 31,2016, Fako Travel Agency has an Accounts Receivable balance of $96,000. Allowance for Doubtful Accounts has a credit balance of $830 before the year-end adjustment. Service revenue...
-
Write a statement that assigns total_coins with the sum of nickel_count and dime_count. Sample output for 100 nickels and 200 dimes is: 300 Learn how our autograder works 542962 3875132.4x3zqy7 1...
-
Beck Corporation has one issue of preferred stock and one issue of common stock outstanding. Given Becks stockholders equity account that follows, determine the original price per share at which the...
-
For a gas described by the Redlich/Kwong equation and for a temperature greater than T c , develop expressions for the two limiting slopes, Note that in the limit as P 0, V , and that in the limit...
-
Show that the density-series second virial coefficients can be derived from isothermal volumetric data via the expression: B = lim(Z 1)/p p-0 p(molar density)= 1/V -
-
Use transaction analysis charts to analyze the following adjustments: a. Depreciation on equipment, $700. b. Rent expired, $300.
-
Suzanne acquired the following ordinary shares in Quarine plc: She made no further acquisitions and the shares were valued at 3.20 each on 31 March 1982. On 24 July 2020, Suzanne sold 1,200 shares...
-
In May 2015, Ruth sold a freehold building which she had used exclusively for business purposes. The building was sold for 220,000, realising a chargeable gain of 42,500. In the following month, Ruth...
-
Select an agreement from Table 19.1 and investigate the details. For additional readings and websites pertaining to the material inthis chapter, see www.mhhe.com/field8e. Name of Agreement Marine...
-
Roseanne is a Scottish taxpayer. She starts trading on 1 July 2020 and prepares accounts for the 16 months to 31 October 2021. The adjusted trading profit for this period is 38,560. Her only other...
-
Joan has been a full-time director of Sunnybank Pursuits Ltd (a trading company) since 2001 and has owned 10% of the company's ordinary shares since 2002. She retired in February 2021 and gave all of...
-
Let x1, x2,. . . , xn be a distinct set of sample points. (a) Prove that the functions f1(x),... , fk(x) are linearly independent if their sample vectors f1......... f are linearly independent...
-
r = 0.18 Find the coefficients of determination and non-determination and explain the meaning of each.
-
For a binary gas mixture described by Kqs, (3,381 and (l l.62l. prove that: See also Eq, (11.87), and note that δ12 = 2B12 - B11 - B22. ds1g d2812 PyIV2 dT
-
What is the ideal work for the separation of an equimolar mixture of methane and ethane at 175C and 3 bar in a steady-flow process into product streams of the pure gases at 35C and 1 bar if Ta = 300...
-
What is the work required for the separation of air (21-mol-% oxygen and 79-mol-% nitrogen) at 25C and 1 bar in a steady-flow process into product streams of pure oxygen and nitrogen, also at 25C and...
-
why you assume value of Machine A is assumed to decrease by $7,000 each year for the purpose of these calculations?
-
1. You want to create a Dog object. Which of the following would you use to describe it? (Choose all that apply.) A. String breed B. int age C. boolean isADog D. String ownerName E....
-
Part 1: Cathy Forth Photography Services (20 marks) You have been hired by Cathy Forth as her new bookkeeper. She has given you the following information: Use the current date and provide an...

Study smarter with the SolutionInn App


