For a gas described by the Redlich/Kwong equation and for a temperature greater than T c ,
Question:
For a gas described by the Redlich/Kwong equation and for a temperature greater than Tc, develop expressions for the two limiting slopes,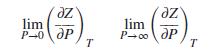 Note that in the limit as P → 0, V → ∞, and that in the limit as P → ∞, V → b.
Note that in the limit as P → 0, V → ∞, and that in the limit as P → ∞, V → b.
Transcribed Image Text:
lim ze, lim P-0dP ӘР P-co dP T (2)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
The RedlichKwong equation of state for a gas is given by P RTVb asqrtT V Vb where P is the pressure ...View the full answer

Answered By

Darwin Romero
I use a hands-on technique and am approachable to my students. I incorporate fun into my lessons when possible. And while my easy-going style is suitable for many subjects and grades, I am also able to adapt my style to the needs of the student. I can describe myself as friendly, enthusiastic and respectful. As a teacher, we can easily get respect from the students if they would feel respected first
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
The ideal gas equation states that P = nRT/V, where P is the pressure, V is the volume, T is the temperature, R = 0.08206 (L atm)/(mol K) is the gas constant, and n is the number of moles. Real...
-
A gas flows at V = 200 m/s, p = 125 kPa, and T = 200C. For (a) Air and (b) Helium, compute the maximum pressure and the maximum velocity attainable by expansion or compression.
-
For a gas whose equation of state is P (v - b) = RT, the specified heat difference cp - cv is equal to (a) R (b) R - b (c) R + b (d) 0 (e) R (1 + v/b)
-
Predicting Genetic disorders in Children based on their Medical information and History Hereditary diseases is becoming more of a problem as human population grows exponentially. Genetic testing...
-
Some sales organizations like to put their sales representatives on a straight commission pay plan which means that they must successfully sell the product in order to receive any money at the end of...
-
The total tax deduction for depreciation is the same over the life of the project regardless of depreciation method. Why then would one be concerned about the depreciation method for capital...
-
Could an include use case have a secondary actor? Explain.
-
Shanes Roasted Peanuts, Inc., supplies snack foods. The business experienced the following events. a. Shanes Roasted Peanuts received cash from the sole stockholder and issued common stock. b. Cash...
-
Discuss the complexities involved in international financial management, including foreign exchange risk management, global capital budgeting, and cross-border investment strategies.
-
Define a new class named UnweightedGraphWithGetPath that extends UnweightedGraph with a new method for finding a path between two vertices with the following header: public List getPath(int u, int...
-
Recreational scuba diving using air is limited to depths of 40 m. Technical divers use different gas mixes at different depths, allowing them to go much deeper. Assuming a lung volume of 6 liters,...
-
Figure 3.3 suggests that the isochores (paths of constant volume) are approximately straight lines on a P-T diagram. Show that the following models imply linear isochores. (a) Constant-, equation...
-
The conditions of Picard's Theorem may fail at a given point for a differential equation, but the equation may still have a unique solution through the point. (In other words, the converse of...
-
Karen bought a house in 1993 for 42,000. In November 1994 she spent 18,000 on dividing the house into two self-contained flats. In September 2018 she sold one of the flats for 95,000, at which time...
-
On 19 April 2019, a close company (which makes up accounts to 31 March annually) lends 100,000 to Siobhan, who is a director of the company and who owns 30% of its ordinary share capital. The company...
-
Consider a small home project or a simple activity to help the climate that you, and maybe some friends, could do. Itemize and estimate the costs, including opportunity costs, of the project or...
-
On 1 March, Joe Conday started a new business. During March he carried out the following transactions: Required: Draw up a statement of financial position for the business at the end of each day. 1...
-
Consider the example of the three homeowners around the lake (the ones depicted in Table 4.2). Suppose the lake was cleaned up to the efficient level of 2 ppm and that the total costs of the cleanup...
-
Suppose f1(t).........fk(t) are vector-valued functions from R to Rn. (a) Prove that if f1(t0),. . . fk(t0) afe linearly independent vectors in Rn at one fixed to, then f1 (t),. . . fk(t) are...
-
Citing a scientific article, explain in your own words, how DNA fingerprinting has been used in forensic science to solve crimes and why it may not always be accurate or effective.
-
Rework Pb. 10.3 for one of the follow ing: (a) i = 65C: (b) t = 75C; (c) t = 85C: (d) t = 95C.
-
Ten (10) kmol hr-1 of hydrogen sulfide gas is burned with the stoichiometric amount of pure oxygen in a special unit. Reactants enter as gases at 25C and 1(atm). Products leave as two streams in...
-
Physiological studies show the neutral comfort level (NCL) of moist air to correspond to an absolute humidity of about 0.01 kg H2O per kg of dry air. (a) What is the vapor-phase mole fraction of H2O...
-
A bank can either invest money for three months at 4.00% or for nine months at 4.50%. Ignoring actual/360 day count adjustments for the purpose of this question, the three against nine FRA quote the...
-
Shuggy Otis, an executive at Slapfish Corp. (SC) intends to retire in 11 years. SC just announced that it will start depositing $500.00 at the end of each quarter into each of its workers' retirement...
-
On January 1, the Hanover Beverage Company replaced the palletizing machine on one of its juice lines. The cost of the machine was $195,000. The machine's expected life is five years or 480,000...

Study smarter with the SolutionInn App


