For an ideal gas prove that: AS c dT V + In, Vo R To R T
Question:
For an ideal gas prove that: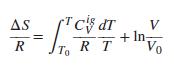
Transcribed Image Text:
AS c dT V + In, Vo R To R T
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
According to first law of thermodynamicsAUqw AU qw qPdV 9 qAU nRT dV fromidea...View the full answer

Answered By

Akash M Rathod
I have been utilized by educators and students alike to provide individualized assistance with everything from grammar and vocabulary to complex problem-solving in various academic subjects. I can provide explanations, examples, and practice exercises tailored to each student's individual needs, helping them to grasp difficult concepts and improve their skills.
My tutoring sessions are interactive and engaging, utilizing a variety of tools and resources to keep learners motivated and focused. Whether a student needs help with homework, test preparation, or simply wants to improve their skills in a particular subject area, I am equipped to provide the support and guidance they need to succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
For an ideal gas flowing through a normal shock, develop a relation for V2/V1 in terms of k, Ma1, and Ma2.
-
For an ideal gas flowing through a normal shock, develop a relation for V2/V1 in terms of k, Ma1, and Ma2.
-
For an ideal gas with constant specific heats show that the compressor and turbine isentropic efficiencies may be written as The states 1 and 2 represent the compressor inlet and exit states and the...
-
In the previous problem, assume the equity increases by 1,500 solaris due to retained earnings. If the exchange rate at the end of the year is 1.24 solaris per dollar, what does the balance sheet...
-
Name the four major steps to prepare for an interview.
-
I dont understand this. Last year [year 1], we decided to drop our highest-end Red model and only produce the Yellow and Green models, because the cost system indicated we were losing money on Red....
-
Define what use case modeling is and is not.
-
The following data show the costs charged by a tax preparation service for a random sample of 15 tax returns. These data can also be found in the Excel file titled tax prep costs. xlsx. a. Using a...
-
Campbell, a single taxpayer, earns $400,000 in taxable income and $2,000 in interest from an investment in State of New York bonds. (Use the U.S. tax rate schedule.) Required: a. How much federal tax...
-
Suppose you want to estimate the effect of Philadelphia's sugar tax, which imposes a 1.5 cent per ounce tax on sugar sweetened beverages. To do so, you collect data on monthly purchases of sugar...
-
A heat engine operating in outer space may be assumed equivalent to a Carnot engine operating between reservoirs at temperatures T H and T C . The only way heat can be discarded from the engine is by...
-
A mass m of liquid water at temperature T 1 is mixed adiabatically and isobarically with an equal mass of liquid water at temperature T 2 . Assuming constant C P , show and prove that this is...
-
If you saw the label phenylephrine . HCl on a decongestant, would you worry that consuming it would expose you to the strong acid hydrochloric acid? Explain. It H . N. CI- CH3...
-
Give a linear-time algorithm for reversing a string.
-
An array is bitonic if it consists of an increasing sequence of keys followed immediately by a decreasing sequence of keys. Given a bitonic array, design a logarithmic algorithm to find the index of...
-
Write a program MooresLaw that takes a command-line argument \(n\) and outputs the increase in processor speed over a decade if microprocessors double every \(n\) months. How much will processor...
-
Write a Queue client that takes an integer command-line argument \(k\) and prints the kth from the last string found on standard input.
-
Given a sorted array of Comparable items, write functions floor () and ceiling () that return the index of the largest (or smallest) item not larger (or smaller) than an argument item in logarithmic...
-
(a) Can you construct an example of a subset S R2 with the property that eve S for any c R, v S, and yet S is not a subspace? (b) What about an example where v + w S for every v, w S, and yet S...
-
Independent random samples of sizes n1 = 30 and n2 = 50 are taken from two normal populations having the means 1 = 78 and 2 = 75 and the variances 21 = 150 and 22 = 200. Use the results of Exercise...
-
A natural-gas fuel contains 85 mol-% methane, 10 mol-% ethane, and 5 mol-% nitrogen. (a) What is the standard heat of combustion (kJ mol-1) of the fuel at 25(C with H2O(g) as a product? (b) The fuel...
-
Calculate the heat capacity of a gas sample from the following information: The sample comes to equilibrium in a flask at 25(C and 121.3 kPa. A stopcock is opened briefly, allowing the pressure to...
-
(a) For one of the compounds listed in Table B.2 of App. B, evaluate the latent heat of vaporization (Hn by Eq. (4.12). How does this result compare with the value listed in Table B.2? (b) Handbook...
-
Some users on your network use Acme Bank for their personal banking. Those users have all recently been the victim of an attack, in which they visited a fake Acme Bank website and their logins were...
-
Solve ADEF. Round to the nearest whole number when necessary. Give angle measures in degrees. 4 E |DE= 4 mZDz DF mZE= 90 EF= 6 m/F 9 LL F
-
Find the total energy available from a rechargeable 1.25-V flashlight battery with a 1.2-Ah rating. If all the energy in a 9-V transistor radio battery rated at 0.392 Ah is used to lift a 150-lb man,...

Study smarter with the SolutionInn App


