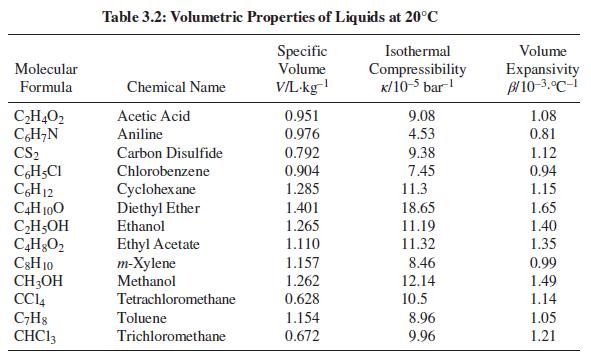
For one of the substances in Table 3.2, compute the change in volume and work done when
Question:
For one of the substances in Table 3.2, compute the change in volume and work done when one kilogram of the substance is heated from 15°C to 25°C at a constant pressure of 1 bar.
Table 3.2
Transcribed Image Text:
Table 3.2: Volumetric Properties of Liquids at 20°C Specific Volume Isothermal Volume Molecular V/L-kg Compressibility K/10-5 bar! Expansivity Bl 10-3.°C-i Formula Chemical Name 1.08 CH,O2 CH;N CS2 Acetic Acid 0.951 0.976 9.08 Aniline 4.53 0.81 0.792 9.38 1.12 0.94 Carbon Disulfide CH,CI CH12 C4H 100 CH5OH Chlorobenzene 0.904 7.45 11.3 Cyclohex ane Diethyl Ether Ethanol 1.285 1.15 1.401 18.65 1.65 1.265 11.19 1.40 Ethyl Acetate т-Хylene Methanol 1.110 11.32 1.35 C3H 10 CH;OH CCI4 C;H8 CHC13 1.157 8.46 0.99 1.262 0.628 12.14 1.49 Tetrachloromethane 10.5 1.14 Toluene 1.154 8.96 1.05 Trichloromethane 0.672 9.96 1.21
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
To calculate the change in volume and work done for heating one kilogram of the substance from 15C t...View the full answer

Answered By

FELIX NYAMBWOGI
I have been tutoring for over 5 years, both in person and online. I have experience tutoring a wide range of subjects, including math, science, English, and history. I have also worked with students of all ages, from elementary school to high school.
In addition, I have received training in effective tutoring strategies and techniques, such as active listening, questioning, and feedback. I am also proficient in using online tutoring platforms, such as Zoom and Google Classroom, to effectively deliver virtual lessons.
Overall, my hands-on experience and proficiency as a tutor has allowed me to effectively support and guide students in achieving their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
For one of the substances in Table 3.2, compute the final pressure when the substance is heated from 15C and 1 bar to 25C at constant volume. Table 3.2 Table 3.2: Volumetric Properties of Liquids at...
-
A piston/cylinder device keeping a constant pressure has 1 kg water at 20oC and 1 kg of water at 100oC both at 500 kPa separated by a thin membrane. The membrane is broken and the water comes to a...
-
A gas is compressed at a constant pressure of 0.800 atm from 9.00 L to 2.00 L. In the process, 400 J of energy leaves the gas by heat. (a) What is the work done on the gas? (b) What is the change in...
-
Models that pertain to the distribution of a resource within supply chains are often referred to as networks. Distribution among these networks is key to the success of a business while also keeping...
-
What is the difference between a programmed and non-programmed decision? Consider a programmed decision and a non-programmed decision you have made recently. Compare and contrast the approaches you...
-
On December 31, 2009, Milo Company had 1,300,000 shares of $5 par common stock issued and outstanding. The stockholders equity accounts at December 31, 2009, had the balances listed here. Common...
-
Why do you think a salesperson might experience call reluctance? How can it be overcome?
-
Comparative financial statement data for Duran Corporation and Kiepert Corporation, two competitors, appear below. All balance sheet data are as of December 31, 2012. Instructions(a) Comment on the...
-
1. Why is steel a good material for cables? 2. What is wrong with a stone suspension bridge? 3. What is reinforced concrete?
-
Four identical SRS arms are grasping a common object as shown below. a. Find the number of degrees of freedom of this system while the grippers hod the object rigidly (no relative motion between the...
-
For one of the substances in Table 3.2, compute the change in volume and work done when one kilogram of the substance is compressed from 1 bar to 100 bar at a constant temperature of 20C. Table 3.2...
-
Various species of hagfish, or slime eels, live on the ocean floor, where they burrow inside other fish, eating them from the inside out and secreting copious amounts of slime. Their skins are widely...
-
If a test that has reliability r is lengthened by a factor n (n ( 1), the reliability R of the new test is given by If the reliability is r0.6, the equation becomes (a) Find R(1). (b) Find R(2); that...
-
Provide a brief discussion of database connection using the JDBC API, which includes: a. Two popular methods used to establish a connection b. Operational procedure to establish a connection c. How...
-
Connect with two classmates to share LinkedIn profiles. Use the criteria in Figure 25 to provide feedback to each other. Figure 25 CRITERIA Name is consistent with cover letter and resume. Audio...
-
A distributed transaction is defined as accessing data source(s) at location(s). a. Single, single b. Multiple, same c. Multiple, different d. Single, multiple
-
In small groups, discuss the following and identify which organizational pattern might work best in each situation. Review Figure 6 for a description and example of each pattern: benefits, issues or...
-
Do you feel more competent preparing content or delivering a presentation? How can you use your skills from one to improve your competence at the other? For example, are you good at organizing a...
-
Let A be an m n matrix. Suppose that C = (A B ) is an m (n + k) matrix whose first n columns are the same as A. Prove that mgC mg A. Thus, appending more columns cannot decrease the size of a...
-
6 (a) Briefly develop a mathematical model of the behaviour of a copper-twisted pair cable (b) Derive the magnetic energy from: w given that: K + w, where the - - k symbols have their usual meaning...
-
Steam at 2,000 kPa containing 6% moisture is heated at constant pressure to 575oC. How much heat is required per kilogram?
-
Steam at 2.700 kPa and with a quality of 0.90 undergoes a reversible, adiabatic expansion in a non-flow process to 400 kPa. It is then healed at constant volume until it is saturated vapor. Determine...
-
Four kilograms of steam in a piston/cytinder device at 400 kPa and 175oC undergoes a mechanically reversible, isothermal compression to a final pressure such that the steam is just saturated....
-
Briefly define TinyOS and explain its goals as an embedded operating system.?
-
Consider the graph shown on the right. Find the strongly connected components of the graph. Whenever you use DFS and there is a choice of nodes to explore, always pick the one that is alphabetically...
-
After viewing the previous video, "Types of Operating Systems As Fast As Possible", explain the difference between embedded operating systems and desktop operating systems. (300-yep, you guessed it)...

Study smarter with the SolutionInn App


