For one of the substances in Table 3.2, compute the final pressure when the substance is heated
Question:
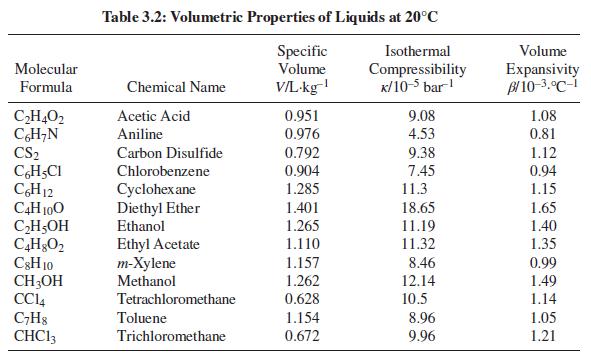
For one of the substances in Table 3.2, compute the final pressure when the substance is heated from 15°C and 1 bar to 25°C at constant volume.
Table 3.2
Transcribed Image Text:
Table 3.2: Volumetric Properties of Liquids at 20°C Specific Volume Isothermal Volume Molecular V/L-kg Compressibility K/10-5 bar! Expansivity Bl 10-3.°C-i Formula Chemical Name 1.08 CH,O2 CH;N CS2 Acetic Acid 0.951 0.976 9.08 Aniline 4.53 0.81 0.792 9.38 1.12 0.94 Carbon Disulfide CH,CI CH12 C4H 100 CH5OH Chlorobenzene 0.904 7.45 11.3 Cyclohex ane Diethyl Ether Ethanol 1.285 1.15 1.401 18.65 1.65 1.265 11.19 1.40 Ethyl Acetate т-Хylene Methanol 1.110 11.32 1.35 C3H 10 CH;OH CCI4 C;H8 CHC13 1.157 8.46 0.99 1.262 0.628 12.14 1.49 Tetrachloromethane 10.5 1.14 Toluene 1.154 8.96 1.05 Trichloromethane 0.672 9.96 1.21
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
To solve this problem we will use the ideal gas law PV nRT where P is the pressure V is the volume n ...View the full answer

Answered By

FELIX NYAMBWOGI
I have been tutoring for over 5 years, both in person and online. I have experience tutoring a wide range of subjects, including math, science, English, and history. I have also worked with students of all ages, from elementary school to high school.
In addition, I have received training in effective tutoring strategies and techniques, such as active listening, questioning, and feedback. I am also proficient in using online tutoring platforms, such as Zoom and Google Classroom, to effectively deliver virtual lessons.
Overall, my hands-on experience and proficiency as a tutor has allowed me to effectively support and guide students in achieving their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
For one of the substances in Table 3.2, compute the change in volume and work done when one kilogram of the substance is compressed from 1 bar to 100 bar at a constant temperature of 20C. Table 3.2...
-
For one of the substances in Table 3.2, compute the change in volume and work done when one kilogram of the substance is heated from 15C to 25C at a constant pressure of 1 bar. Table 3.2 Table 3.2:...
-
The following standard costs were developed for one of the products of ABC Company: STANDARD COST CARD PER UNIT Direct materials: 20 pounds x $10 per pound .................................. $200.00...
-
Calculate the weighted average cost of capital for Genedak-Hogan for before and after international diversification. Did the reduction in debt costs reduce the firm's weighted average cost of...
-
1. How is New Belgium Brewing Companys approach to social responsibility different from that of other companies? What are the advantages and disadvantages of this approach? New Belgium is a small,...
-
Consider the following information on Stock I and II: The market risk premium is 12 percent, and the risk-free rate is 4 percent. Which stock has the most systematic risk? Which one has the most...
-
How would you distinguish objects from classes? Provide an example. a. Comment on the statement: A class is an abstraction of objects. b. Comment on the statement: A class is a template for creating...
-
A city established a public housing authority to fund the construction of low-income residential homes within city limits. The authority is governed by a nine-person board of trustees. New trustees...
-
What role does strategic communication play in crisis mitigation and reputation management, leveraging narrative frameworks to maintain stakeholder trust and confidence?
-
A wholesale grocery distribution center uses a two-step process to fill orders. Tomorrows work will consist of filling the seven orders shown. Determine a job sequence that will minimize the time...
-
a. A vacant lot acquired for $115,000 is sold for $298,000 in cash. What is the effect of the sale on the total amount of the sellers (1) assets, (2) liabilities, and (3) owners equity? b. Assume...
-
A substance for which is a constant undergoes an isothermal, mechanically reversible process from initial state (P 1 , V 1 ) to final state (P 2 , V 2 ), where V is molar volume. (a) Starting with...
-
A man is rowing at 8 km/h in a river 1.5 km wide in which the current is 5 km/h. (a) In what direction should he head in order to get across the river in the shortest possible time? (b) How much time...
-
Telford Engineers plc, a medium-sized manufacturer of automobile components, has decided to modernise its factory by introducing a number of robots. These will cost 20 million and will reduce...
-
Saeed made the following purchases of ordinary shares in Hyban plc: In June 2017, the company made a 1 for 20 rights issue at 1.50 per share and Saeed took up the shares which he was offered....
-
Boswell Enterprises Ltd is reviewing its trade credit policy. The business, which sells all of its goods on credit, has estimated that sales revenue for the forthcoming year will be 3 million under...
-
Virtual randomization requires the timezero average for feral hosts to be the same as that for giant runts, but the temporal trends are otherwise unconstrained. It appears that the model matrix...
-
Following are some illustrative numbers for benefits and costs arising from a program to restrict emissions of a pollutant. Current emissions are 10 tons per month. Identify the emission level at...
-
(a) Prove that 1 +t2,1 +t2,1 + 2t +t2 is a basis for the space of quadratic polynomials P(2). (b) Find the coordinates of p(t) = 1 + 4t + 7t2 in this basis.
-
In each of the following independent cases, document the system using whatever technique(s) your instructor specifies. a. Dreambox Creations (www.dreamboxcreations.com/) in Diamond Bar, California,...
-
For the acetone (1)/methanol(2) system a vapor mixture for which z1 = 0,25 and z2 = 0.75 is cooled to temperature T in the two-phase region and flow* into a separation chamber at a pressure of 1 bar....
-
A process stream contains light species 1 and heavy species 2. A relatively pure liquid stream containing mostly 2 is desired, obtained by a single-stage liquid/vapor separation. Specifications on...
-
Assuming the validity of the De Priester charts, make the following VLE calculations for the methane(1)/cthylenc(2)/elhand(3) system: (a) BVBL P. given x1 = 0.10. x2 = 0.50, and t = -60(F). (b) DEW...
-
Bronchial alveolar lavage and four endobronchial biopsies were performed and submitted to the insurance carrier as follows: 31625 x 4 units, 31624-51. Is this claim coded appropriately?
-
What goals are used to measure labor and management partnerships?
-
1. A Supervisor email you a question. 2. Potential client comes to your office. He has $40000 in wages on W-2. He also listed $30000 of charitable contributions, mostly clothing, on a single piece of...

Study smarter with the SolutionInn App


