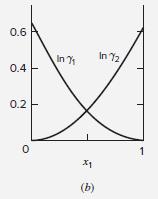
Table 13.10 gives values of parameters for the Wilson equation for the acetone(1)/methanol(2) system. Estimate values of
Question:
Table 13.10 gives values of parameters for the Wilson equation for the acetone(1)/methanol(2) system. Estimate values of ln γ∞1 and ln γ∞2 at 50°C. Compare with the values suggested by Fig. 13.4(b). Repeat the exercise with the NRTL equation.
Table 13.10
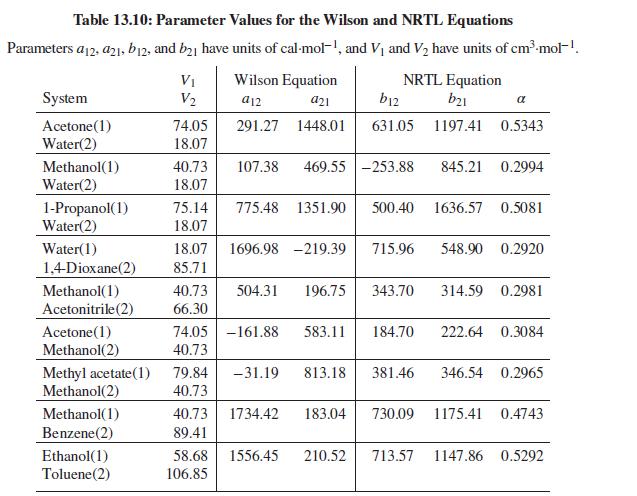
Fig. 13.4(b)
Transcribed Image Text:
Table 13.10: Parameter Values for the Wilson and NRTL Equations Parameters a12, a21, b12, and ba1 have units of cal-mol-!, and Vị and V2 have units of cm mol-!. Wilson Equation NRTL Equation b12 VI System V2 a12 b21 a 74.05 18.07 Acetone(1) 291.27 1448.01 631.05 1197.41 0.5343 Water(2) 469.55 -253.88 Methanol(1) Water(2) 40.73 107.38 845.21 0.2994 18.07 75.14 775.48 1351.90 1-Propanol(1) Water(2) 500.40 1636.57 0.5081 18.07 Water(1) 18.07 1696.98 -219.39 715.96 548.90 0.2920 1,4-Dioxane(2) 85.71 Methanol(1) 40.73 504.31 196.75 343.70 314.59 0.2981 Acetonitrile(2) 66.30 Acetone(1) 74.05 -161.88 583.11 184.70 222.64 0.3084 Methanol(2) Methyl acetate(1) Methanol(2) 40.73 79.84 -31.19 813.18 381.46 346.54 0.2965 40.73 Methanol(1) 40.73 1734.42 183.04 730.09 1175.41 0.4743 Benzene(2) 89.41 210.52 Ethanol(1) Toluene(2) 58.68 1556.45 713.57 1147.86 0.5292 106.85
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
To estimate the values of ln and ln for the acetone1methanol2 system at 50C using the Wilson and NRTL equations we can use the parameters given in Tab...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
Repeat Exercise 13 for the integral In Exercise 13 Determine the values of n and h required to approximate To within 105 and compute the approximation. Use a. Composite Trapezoidal rule. b. Composite...
-
Table B.2 of Appendix B provides parameters for an equation that gives P sat as a function of T for a number of pure compounds. For one of them, determine the heat of vaporization at its normal...
-
Repeat Exercise 1 using four-digit rounding arithmetic, and compare the errors to those in Exercise 3. In Exercise 1 a. b. 0.5 0.4794 0.6 0.5646 0.7 0.6442 x f(x) f'(x) 0.0 0.00000 0.2 0.74140 0.4...
-
How much taxes do Employees and Employers pay in F.I.C.A? a) Employees and Employers paid 1.45% in taxes. b) Employees and Employers paid 7.65% in taxes. c) Employers and Employees paid 2.7% in...
-
Explain how both an emission tax and tradable pollution permits system can reduce pollution.
-
Sorbo Company is planning to add a new product to its line. To manufacture this product, the company needs to buy a new machine at a $600,000 cost with an expected four-year life and a $20,000...
-
Managers must be problem-solvers but are not always decision-makers. Do you agree? Disagree? Provide an example that clarifies your position.
-
In a statement to Gillettes shareholders, Chairman and CEO James Kilts indicated, Despite several new product launches, Gillettes advertising-to-sales declined dramatically . . . to 6.5 percent last...
-
Concord Corporation began operations in July 2022. At the end of the month, the company prepares monthly financial statements. It has the following information for the month. 1. At July 31, the...
-
A marketer of health foods is attempting to segment a certain market on the basis of consumer self-image. Describe the four types of consumer self-image and discuss which one(s) would be most...
-
Consider the following model for G E RT of a binary mixture: This equation in fact represents a family of two-parameter expressions for G E RT; specification of k leaves A 12 and A 21 as the free...
-
Possible correlating equations for ln 1 in a binary liquid system are given here. For one of these cases, determine by integration of the Gibbs/Duhem equation [Eq. (13.11)] the corresponding...
-
The accompanying diagram depicts a monopolist whose price is regulated at $10 per unit. Use this figure to answer the questions that follow. a. What price will an unregulated monopoly charge?b. What...
-
Which viewpoint emphasizes that business can find ways to profit by doing things that advance the well-being of society? (a) classical (b) shared value (c) defensive (d) obstructionist
-
Pluralism and the absence of discrimination and prejudice in policies and practices are two important hallmarks of _________. (a) the glass ceiling effect (b) a multicultural organization (c) quality...
-
Trends in the new workplace include which of the following? (a) More emphasis by managers on giving orders. (b) More attention by organizations to valuing people as human assets. (c) Less teamwork....
-
Kim Davidovic owns and operates a small retail store selling the outdoor clothing of a Canadian manufacturer to a predominately university-student market. Lately, a large department store outside of...
-
What is a tax preference item?
-
For the following exchange matrices, find nonnegative price vectors that satisfy the equilibrium condition 3. (a) (b) (c) 1-3 2-3 1-2 1-2 1212 0 001 1-2 1-3 1-6 35 .50 30 25 .20 .30 40 30 40
-
What are current assets and current liabilities? How are they different from non-current assets and non-current liabilities?
-
The virial coefficient can be related to the pair potential by Eqn. 7.59. (a) Derive the integrated expression for the second virial coefficient in terms of the square well potential parameters /k, ,...
-
What forms does the derivative (C V /V) T have for a van der Waals gas and a Redlich- Kwong gas? Comment on the results.
-
Develop an expression for the Gibbs energy departure function based on the Redlich- Kwong (1958) equation of state: Z = 1 + bp ap 1-bp RT/(1+bp)
-
How does the concept of intersectionality, as developed by Kimberl Crenshaw, challenge traditional understandings of identity and social categorization by highlighting the interconnectedness and...
-
To what extent does intersectional scholarship inform policy-making and advocacy efforts aimed at addressing social disparities and promoting equity and justice for marginalized communities ?
-
How can intersectionality contribute to building solidarity and fostering coalitions across diverse social movements, while also recognizing and respecting the distinctiveness of different...

Study smarter with the SolutionInn App


