At what temperature does water boil on the top of Mount Everest, elevation z = 8848 m?
Question:
At what temperature does water boil on the top of Mount Everest, elevation z = 8848 m?
Recall that the dependence of pressure with altitude is given by: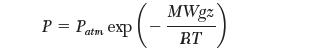
where, Patm is atmospheric pressure, g is the gravitational acceleration, and MW is the molecular weight of the gas.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:





