Calculate the van der Waals parameter a for CH4, C6H6, and CH3OH. Based on these values, estimate
Question:
Calculate the van der Waals parameter a for CH4, C6H6, and CH3OH. Based on these values, estimate the value of C6 for each species. Compare the values obtained with that calculated by Equation (4.8).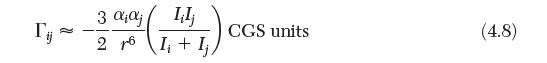
Transcribed Image Text:
3 ;j Lilj 26 I+Ij CGS units a (4.8)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Calculate the van der Waals parameter b for CH4, C6H6, and CH3OH. Based on these values, estimate the molecular diameter of each species. Compare the values obtained with those in Table 4.2. TABLE...
-
Van der Waals Equation and Critical Points (a) In p V- diagrams the slope p/V along an isotherm is never positive. Explain why. (b) Regions where p/V = 0 represent equilibrium between two phases;...
-
Calculate the van der Waals parameters of carbon dioxide from the values of the critical constants and compare your results with the values for and b in Table 7.4. Table 7.4 RedlichKwong van der...
-
Let F = [F] lb. a) Determine the force Fp acting at roller D. b) Determine the force FE acting at pin E. F 4 ft B 600 lb. ft -3 ft C -3 ft OD -2 ft- E
-
Analyze the debit and credit aspect of each transaction listed at (a), (b) and (c) of question B3.2? (a) Owner puts cash into the business. (b) Buy a vehicle for cash. (c) Receive a bill for...
-
After noticing that Jennifers baby was not eating well and was small for his age, doctors tested him for drugs. When the test results proved positive, Jennifer admitted to police that she had...
-
Air flows though the mitered bend shown in Fig. P8.57 at a rate of \(5.0 \mathrm{cfs}\). To help straighten the flow after the bend, a set of 0.25-in.-diameter drinking straws is placed in the pipe...
-
Determining the cash flow annuity with income tax considerations To open a new store, Alpha Tire Company plans to invest $240,000 in equipment expected to have a four-year useful life and no salvage...
-
2. Set up the following equations in the computer form: a. x+y=2+y-z 3z-1 b. 2xzy+x+ 4(3z+1)-y C. x = (y + z)
-
Consider a cylinder fi tted with a piston that contains 2 mol of H2O in a container at 1000 K. Calculate how much work is required to isothermally and reversibly compress this gas from 10 L to 1 L,...
-
Using data from Table 4.2, estimate the equilibrium bond length that would exist in a molecule of Xe2. TABLE 4.2 Lennard-Jones Parameters for Several Species /k(K) 10.2 35.7 Gas He H CH4 C6H6 288 28...
-
Why does the machining cost increase rapidly as surface finish requirements become finer?
-
What strategies can multinational corporations employ to effectively manage diverse cultural, political, and economic factors across various geographic regions while maintaining a cohesive global...
-
Superior Micro Products uses the weighted-average method in its process costing system. Data for the Assembly Department for May appear below: Work in process, May 1 Cost added during May Equivalent...
-
What organizational design would be most appropriate for a sporting goods manufacturer? For a sport marketing agency? For an organization bidding for the right to host a major international event?...
-
You are planning security protocols for your companys new server room. Whats the sim- plest way to help physically keep potential attackers away from your servers?
-
Definition of Open strategy Definition of Close strategy Comparison between open strategy and close strategy Examples of companies using Open strategy Implementation of open strategy Evaluation...
-
In each of the following independent situations, an example is given requiring a trade-off between the qualitative characteristics discussed in the text. For each situation, identify the relevant...
-
For what reason might an exporter use standard international trade documentation (letter of credit, draft, order bill of lading) on an intrafirm export to its parent or sister subsidiary?
-
Outline a reasonable mechanism for the formation of tert-butyl methyl ether according to the preceding equation.
-
Write equations describing two different ways in which benzyl ethyl ether could be prepared by a Williamson ether synthesis.
-
Only one combination of alkyl halide and alkoxide is appropriate for the preparation of each of the following ethers by the Williamson ether synthesis. What is the correct combination in each case?
-
Diversity means to me that you have different cultures, religions or skin tones surrounding you. That it's not just one kind of person putting out ideas out there that it varies with different...
-
Please post a main posting addressing the below questions no later than midnight on Day 3 of the week (Wednesday), and then respond to at least 2 other students' postings no later than Day 7 (Sunday)...
-
Please post a main posting addressing the below questions no later than midnight on Day 3 of the week (Wednesday), and then respond to at least 2 other students' postings no later than Day 7 (Sunday)...

Study smarter with the SolutionInn App


