Consider a mixture of 1-propanol (a) and water (b). At 25C, the three-suffi x Margules equation parameters
Question:
Consider a mixture of 1-propanol
(a) and water (b). At 25°C, the three-suffi x Margules equation parameters are: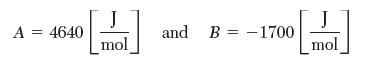
Does this binary mixture form an azeotrope at 25°C? If so, at what pressure does the azeotrope form?
Transcribed Image Text:
A = 4640 J mol and B-1700 J mol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)

Answered By

Krishnavendra Y
I am a self motivated financial professional knowledgeable in; preparation of financial reports, reconciling and managing accounts, maintaining cash flows, budgets, among other financial reports. I possess strong analytical skills with high attention to detail and accuracy. I am able to act quickly and effectively when dealing with challenging situations. I have the ability to form positive relationships with colleagues and I believe that team work is great key to performance. I always deliver quality, detailed, original (0% plagiarism), well-researched and critically analyzed papers.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
Let r and s be solutions to the quadratic equation x 2 b x + c = 0. For n N, define d0 = 0 d1 = r s dn = b dn1 c dn2 (n 2) Prove that dn = r n s n for all n N. [4 marks] (b) Recall that a commutative...
-
(a) LEP Table 12-2: Exothermic Reaction with Heat Exchange Download the Polymath, MATLAB, Python, or Wolfram codes for the algorithm and data given in Table T12-2 for the exothermic gas phase...
-
In Exercises 5 and 6, the transition matrix P for a Markov chain with states 0 and 1 is given. Assume that in each case the chain starts in state 0 at time n = 0. Find the probability that the chain...
-
June Devlin operates a Water Bottling Service in Yellowknife. All transactions requiring the payment of cash are recorded in the cash payments journal (page 14). The account balances as of May 1,...
-
A hollow cylinder is closed at the top end by a movable piston of mass \(M\) that can slide in the hollow part of the cylinder without friction. A flow of gas equal to \(\Phi=\frac{\Delta M_{\text...
-
Radiation interactions are of importance in crystal pullers for growing crystals from melts since these operate at very high temperatures. The radiation view factors change at various stages of...
-
What about your own values when it comes to balancing your home and work life? Assume that youre about to graduate from law school and about to get married to a fiance (e) whos also about to graduate...
-
The following data pertain to ABC Company (assume a perpetual inventory system) for the month ended January 31, 2019: Date Description Units Unit Cost Unit Selling Price Jan.1 Beg. Inventory 5...
-
At 20C and 0.073 bar, a binary liquid mixture of cyclohexane (1) and toluene (2) is in vapor liquid equilibrium. The liquid mole fraction of cyclohexane is measured to be x1 = 0.471. Assume that the...
-
A binary mixture of water (1) and benzene (2) is in vaporliquid equilibrium at a liquid mole fraction x1 = 0.6 and a pressure of 74.5 kPa. The liquid phase nonideality can be described by the...
-
What are four primary sources of law in the United States?
-
The Baseball data set (in Appendix A and Connect) reports information on the 30 Major League Baseball (MLB) teams for the 2021 season. 1. In the MLB, each team plays 162 games in a season. A...
-
Subset the data to select only those individuals who lived in an urban area. Reduce the dimensionality of the data by converting numerical variables such as age, height, weight, number of years of...
-
Ricardian equivalence and fiscal policy First consider an economy in which Ricardian equivalence does not hold. a. Suppose the government starts with a balanced budget. Then, there is an increase in...
-
Subset the data to include only the employees who work in the hardware division and have a college degree. Reduce the dimensionality of the data by converting numerical variables such as age, years...
-
An airline company is going through a major cost reduction effort that involves reducing the number of flights by 20%, increasing fuel efficiency by 5%, and offering to 10% of the pilots and flight...
-
Cost-volume-profit relationship Feskin Corporation is a manufacturing company that makes small electric motors it sells for $36 per unit. The variable costs of production are $22 per motor, and...
-
9.Consider the reaction 3NO2(g)+H2O=2HNO3(aq)+NO(g) where Delta H=-137 kJ.How many kilojoules are released when 92.3g of NO2 reacts?
-
Calculate the final pressure of a sample of water vapour that expands reversibly and adiabatically from 87.3 Torr and 500 dm3 to a final volume of 3.0 dm3. Take Y = 1.3
-
When 178J of energy is supplied as heat to 1.9 mol of gas molecules, the temperature of the sample increases by 1.78 K. Calculate the molar heat capacities at constant volume and constant pressure of...
-
When 2.0 mol CO2 is heated at a constant pressure of 1.25 atm, its temperature increases from 250 K to 277 K. Given that the molar heat capacity of CO2 at constant pressure is 37.11 J K-1 mol-1,...
-
Raider sailboats demand is listed below. The forcast for year 5 is 5 , 6 0 0 sailboats, what will be the demand level for spring of year 5 ?
-
Tim has to wear more than one kind of protective eyewear for his job. What are the requirements in this case?
-
7) The XYZ Company has estimated expected cash flows for 1996 to be as follows: Probability Cash flow .10 $120,000 .15 140,000 .50 150,000 .15 180,000 .10 210,000 Calculate: a. expected value b....

Study smarter with the SolutionInn App


