In an actual expansion through the turbine of Example 3.12, 22.1 [MW] of power is obtained. What
Question:
In an actual expansion through the turbine of Example 3.12, 22.1 [MW] of power is obtained. What is the isentropic effi ciency, hturbine, for the process? The isentropic effi ciency is given by: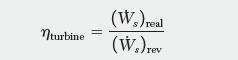

Transcribed Image Text:
nturbine = (W) real (W) rev
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
We run into several different definitions of efficiency depending on the context Isentropic efficien...View the full answer

Answered By

Chiranjib Thakur
I have no tutoring experience yet, but I can share my skills and knowledge gained from my education and work experiences. I have been a CPA since 2012 with 6 years of work experience in internal auditing and 4 years of work experience in accounting at the supervisory level.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Percy is an electrician and works for Cables SA Company. Two months ago, Percy was on his way to work when a bus crashed into his vehicle. Percy sustained severe injuries to his right arm and leg and...
-
Image transcription text Module 5 Discussion A' Instructions: This discussion will be completed in two parts, and will give you an opportunity to reect upon this week's content and to interact with...
-
The vortex tube (also known as a Ranque or Hirsch tube) is a device that produces a refrigeration effect by expanding pressurized gas such as air in a tube (instead of a turbine as in the reversed...
-
A person who is 6 feet and 3 inches tall and weighs 185 pound force (lbf) is driving a car at a speed of 65 miles per hour over a distance of 25 miles between two cities. The outside air temperature...
-
Explain what is meant by 'net assets'?
-
In shooting a free throw in basketball, what is the primary advantage that a high, arching shot has over one with a flatter trajectory? Explain.
-
For oil \(\left(S G=0.86, \mu=0.025 \mathrm{Ns} / \mathrm{m}^{2} ight.\) ) flow of \(0.2 \mathrm{~m}^{3} / \mathrm{s}\) through a round pipe with diameter of \(500 \mathrm{~mm}\), determine the...
-
Naranjo Company designs industrial prototypes for outside companies. Budgeted overhead for the year was $260,000, and budgeted direct labor hours were 20,000. The average wage rate for direct labor...
-
The capital structure of Cain is presented below: Debt @ 10% Common stock Total Common shares Cain $140,000 280,000 $420,000 28,000 Compute the stock price for Cain Auto Supplies if it sells at 17...
-
Steam enters the turbine in a power plant at 600C and 10 MPa and is condensed at a pressure of 100 kPa. Assume the plant can be treated as an ideal Rankine cycle. Determine the power produced per kg...
-
An ideal gas enters a turbine with a fl ow rate of 250 mol/s at a pressure of 125 bar and a specifi c volume of 500 cm3/mol. The gas exits at 8 bar. The process operates at steady-state. Assume the...
-
1. What suggestions would you give Jim to help Airspace lower its operating costs? 2. How would you help Airspace implement those plans? 3. What constraints can you identify that would prevent...
-
A football is launched from a cannon aimed at angles of 7 5 degrees, 6 0 degrees, and 4 5 degrees. For which angle does the football travel the farthest, if there is no air resistance and the speed...
-
Suppose you performed a similar experiment using diphenyl ether as your solvent. Given the data in the table, what would be the freezing point temperature of the solution? Data Result molality of...
-
you are embarking on a cross country roadtrip. when you leave your house on a 35.0 degree celsius day, the pressure in your tires is 2.50 * 5 n/m. what is the pressure in the tires when you reach...
-
Boulevard Industries has decided to provide safety training to its 1,500+ employees who work in the Boulevard main office. Part of this training is a 60-minute presentation by a member of the...
-
A doctor orders gentamicin 4 mg/kg/day in two equal doses IVPB to infuse over 90 minutes for a patient weighing 60 kg. The medication is supplied in 60 mg/100 mL solution. a. How many milligrams will...
-
(a) What role does the EITF play in establishing accounting standards? (b) Why can it meet this role more efficiently than the FASB?
-
How can a promoter avoid personal liability for pre-incorporation contracts?
-
What is the absolute configuration (R or S) of L-glycerol 3- phosphate? What must be the absolute configuration of the naturally occurring phosphatidic acids biosynthesized from it?
-
Spermaceti is a wax obtained from the sperm whale. It contains, among other materials, an ester known as cetyl palmitate, which is used as an emollient in a number of soaps and cosmetics. The...
-
Arachidonic acid is the biosynthetic precursor to PGE2. The structures of PGE1 (see Figure 26.5) and PGE2 are identical except that PGE2 has one more double bond than PGE1. Suggest a reasonable...
-
Emily Company uses a periodic inventory system. At the end of the annual accounting period, December 31 of the c accounting records provided the following information for product 2 Inventory,...
-
Discuss the mechanisms underlying signal transduction in cellular communication, particularly focusing on the intricate interplay between ligands, receptors, and intracellular signaling cascades .
-
At the beginning of Year 2, the Redd Company had the following balances in its accounts: Cash Inventory $8,400 2,400 Common stock 7,900 Retained earnings 2,900 During Year 2, the company experienced...

Study smarter with the SolutionInn App


