You are using the PengRobinson equation of state to determine the entropy change of an ideal gas:
Question:
You are using the Peng–Robinson equation of state to determine the entropy change of an ideal gas: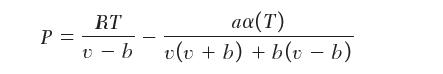
Is it better to try s = s (T,v) or s = s (T,P)? Explain.
Transcribed Image Text:
P = RT v-b aa(T) v(v + b) + b(v - b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)

Answered By

Akshay Singla
as a qualified engineering expert i am able to offer you my extensive knowledge with real solutions in regards to planning and practices in this field. i am able to assist you from the beginning of your projects, quizzes, exams, reports, etc. i provide detailed and accurate solutions.
i have solved many difficult problems and their results are extremely good and satisfactory.
i am an expert who can provide assistance in task of all topics from basic level to advance research level. i am working as a part time lecturer at university level in renowned institute. i usually design the coursework in my specified topics. i have an experience of more than 5 years in research.
i have been awarded with the state awards in doing research in the fields of science and technology.
recently i have built the prototype of a plane which is carefully made after analyzing all the laws and principles involved in flying and its function.
1. bachelor of technology in mechanical engineering from indian institute of technology (iit)
2. award of excellence in completing course in autocad, engineering drawing, report writing, etc
4.70+
48+ Reviews
56+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-6. On December 12, Irene purchased the building where her store is located. She paid...
-
Apple business is currently operating in the country of China and you were asked to report on different aspects of the business as you consider international expansion. By now, you should have a good...
-
On 01/01/2019 ABC Company purchase a truck for $140,000. The truck has a $50,000 salvage value and a life of 4 years with estimated total miles of 200,000. Fill in the answers below: 1. How much is...
-
A company had average total assets of $500,000, gross sales of $575,000, and net sales of $550,000. The companys total asset turnover is a. 1.15. b. 1.10. c. 0.91. d. 0.87. e. 1.05.
-
Choose three different presidents from Harry S. Truman through Barack Obama and look at the data. www.presidency.ucsb.edu/data/popularity.php?pres=44 For this, navigate to website's collection of job...
-
Lynette is the CEO of publicly traded TTT Corporation and earns a salary of $200,000 in the current year. Assume TTT is profitable.What is TTT Corporations after-tax cost of paying Lynettes salary...
-
A trial weight should generate a force equal to 10% of the rotor weight in order to establish the location and amount of imbalance. The rotor weighs 100 lb and has an operating speed of 3,600 rpm....
-
On May 1, Burns Corporation acquired 100 percent of the outstanding ownership shares of Quigley Corporation in exchange for $710,000 cash. At the acquisition date, Quigley's book and fair values were...
-
On September 25, 2023, Homeowner met with Contractor to discuss an addition that Homeowner wanted to add to his house. Homeowner wanted Contractor to build a large primary bedroom with a large,...
-
Consider a gas that undergoes a process from state 1 to state 2. You know the ideal gas heat capacity and an equation of state. Which of the following hypothetical paths would be most appropriate to...
-
Of the following mixture, which do you think has entropy departure function of larger magnitude (a) 50 mol% methane mixed with 50 mol% ethane (b) 50 mol% acetone mixed with 50 mol% chloroform?...
-
Approximately 20 million Americans work for themselves. Most run single-person businesses out of their homes. One-quarter of these individuals use personal computers in their businesses. A market...
-
In particular, you were introduced to three decision-making techniques -- majority rule, minority rule by authority or expertise, and consensus rule -- each with their own pros and cons. Using these...
-
Identify and describe the structures and functions of the cerebral cortex, including the four lobes and two hemispheres.
-
Find the least common denominator (LCD) of the rational expressions: 12 5 (a) 99x7 9x2 2 1 (b) 3x 12' 10x 30
-
Find the yields as of Mar 27on the following Treasury securities: 4-week, 13-week, 26-week, 52-week, 2-year, 3-year, 5-year, 10-year, 20-year, 30-year. Plot a yield curve. Comment on the shape of the...
-
Marie's Magic Shoppe provides the following information about its single product. Targeted operating income $38,000 Selling price per unit $25.00 Variable product cost per unit $6.00 Variable...
-
Give an original example of a dangling or misplaced modifier. Why are introductory verbal phrases dangerous?
-
The Taylor's series expansion for cosx about x = 0 is given by: where x is in radians. Write a user-defined function that determines cosx using Taylor's series expansion. For function name and...
-
The rate of the reaction A + 3 B --7 C + 2 D was reported as 1.0 mol dm-1 S-1. State the rates of formation and consumption of the participants.
-
The rate of consumption of B in the reaction A + 3 B 7 C + 2 D is 1.0 mol dm3 S-1. State the reaction rate, and the rates of formation or consumption of A, C, and D.
-
The rate law for the reaction in Exercise 22.1b was found to be v = k[A][BF, What are the units of k? Express the rate law in terms of the rates of formation and consumption of (a) A, (b) C.
-
calculate the WACC from the data below. The tax rate is 21%. risk free rate (10-year Treasury Yield) 4.99% equity beta 0.52 expected market risk premium 4.00% Based on the historical approach, we...
-
Annual Net Income # shares outstanding Stock price per share Estee Lauder (EL) $3,360,000 231,800 $190.58 Lululemon (LULU) $1,002,000 122,440 $345.32 Louis Vuitton (LVMHF) Paris exchange $13,270,000...
-
What is the "Fischer equation" and what use would this equation have for capital budgeting? Typically, annual depreciation allowances are not indexed to inflation. What is the impact of inflation on...

Study smarter with the SolutionInn App


