According to the following diagram of an H 2 molecule, how many more positivenegative attractions are there
Question:
According to the following diagram of an H2 molecule, how many more positive–negative attractions are there relative to the case of two isolated hydrogen atoms? What do we call this extra positive–negative attraction?
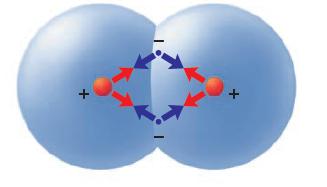
Transcribed Image Text:
+ +
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
There is one more positivenegative attraction in an H2 molecule relative to the case of tw...View the full answer

Answered By

Larlyu mosoti
I am a professional writer willing to do several tasks free from plagiarism, grammatical errors and submit them in time. I love to do academic writing and client satisfaction is my priority. I am skilled in writing formats APA, MLA, Chicago, and Harvard I am a statistics scientist and I can help out in analyzing your data. I am okay with SPSS, EVIEWS, MS excel, and STATA data analyzing tools.
Statistical techniques: I can do linear regression, time series analysis, logistic regression, and some basic statistical calculations like probability distributions. . I'm ready for your working projects!
Services I would offer:
• Academic writing.
• Article writing.
• Data entry.
• PDF conversion.
• Word conversion
• Proofreading.
• Rewriting.
• Data analyzing.
The best reason to hire me:
- Professional and Unique work in writing.
- 100% satisfaction Guaranteed
- within required time Express delivery
- My work is plagiarism Free
- Great communication
My passion is to write vibrantly with dedication. I am loyal and confident to give my support to every client. Because Client satisfaction is much more important to me than the payment amount. A healthy client-contractor relationship benefits in the longer term. Simply inbox me if you want clean work.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
The rates of many atmospheric reactions are accelerated by the absorption of light by one of the reactants. For example, consider the reaction between methane and chlorine to produce methyl chloride...
-
Hydrogen fluoride decomposes according to the following equation The value of Kc at room temperature is 1.0 1095. From the magnitude of Kc, do you think the decomposition occurs to any great extent...
-
Identify the process evaluation article that you chose and explain why you selected this example. Describe the purpose of the evaluation, the informants, the questions asked, and the results of the...
-
Consider the valve failure data in Example 7.6. Set up a one-sided CUSUM chart for monitoring and detecting an increase in failure rate of the valve. Assume that the target value of the mean time...
-
Marcia Young earns $25 per hour for up to 400 units of production per day. If she produces more than 400 units per day, she will receive an additional piece rate of $0.50 per unit. Assume that her...
-
From the following T-accounts, record the following: (a) The July 3 payment for FICA (OASDI and Medicare) and federal income taxes, (b) The July 30 payment of SUTA tax, (c) The July 30 deposit of any...
-
Plaintiff purchased stain and paint from defendant that, upon application, presented significant issues: discoloration and cracking. These issues became apparent 30 days after receipt of the...
-
Mann Hardware has four employees who are paid on an hourly basis plus time-and-a-half for all hours worked in excess of 40 a week. Payroll data for the week ended March 15, 2014, are presented below....
-
Koala Pty Ltd makes premium fishing rods and sells them to fishing retailers around Australia. Their products are in such high demand that Koala Pty Ltd sells everything they make (that is, they have...
-
The polyatomic ion MnO 4 2 is listed in your book as permanganate. What would be the name of MnO 3 2 ?
-
Using only the periodic table, determine which bond in each pair is more ionic: (a) HF or HCl (b) OF or CF
-
Assume that a time-domain sequence generated by using a sampling interval equal to 0.01 is given by \(x(k)=\{0,2,5,12,5,3,3,-1,1,0\}\). Decimate this sequence so that the sampling interval is 0.02 .
-
If you are in charge of a private firm and it doesnt have a share price, what should be your goal as a financial manager? Explain.
-
In 2014 it was reported that Apple had twice as much cash as the US government. Why could this be a problem for the firm?
-
Assume that instead of fully financing the expansion with debt, the managers of ABB Ltd say they wish to maintain the ratio of non-current liabilities to equity after the expansion. What would ABBs...
-
You have been manager of a small company for 20 years and have become great friends with your employees. In the last month, foreign owners have bought out the companys founding owner and have told...
-
Mallory plc has two different bonds currently outstanding. Bond M has a face value of 20,000 and matures in 20 years. The bond makes no payments for the first 6 years, then pays 1,200 every 6 months...
-
This problem takes you through the accounting for sales, receivables, and uncollectibles for Quick Mail Corp., the overnight shipper. By selling on credit, the company cannot expect to collect 100%...
-
Perform the indicated operations. In designing a cam for a fire engine pump, the expression is used. Simplify this expression. (3) (3 4 32
-
Using toluene and acetylene as your only sources of carbon atoms, show how you would prepare the following compound.
-
Using the expression dS = C p /T dt VdP, calculate the decrease in temperature that occurs if 2.25 moles of water at 310. K and 1650. bar is brought to a final pressure of 1.30 bar in a reversible...
-
3.75 moles of an ideal gas with C V ,m = 3/2 R undergoes the transformations described in the following list from an initial state described by T = 298 K and P = 4.50 bar. Calculate q, w, U, H, and S...
-
When tasked with assigning work, a project manager must often consider whether tasks should be performed by a group of workers or by individual workers. How should a project manager decide which is...
-
Prepare a brief summary of the case study and the issues presented. b. Identify and describe the issues or challenges presented in the case. c. Identify six (6) principles/concepts and explain each...
-
Amazon are both companies with world-class logistics systems in place. Walmart does most of their own logistics and Amazon hires out a lot of it. How are both of these organizations able to be...

Study smarter with the SolutionInn App


