Consider the organic compound C 3 H 6 O 2 , methyl acetate. (a) What is the
Question:
Consider the organic compound C3H6O2, methyl acetate.
(a) What is the problem with using the shortcut method to assign oxidation numbers to the atoms in methyl acetate?
(b) The dot diagram of methyl acetate is shown below. Assign an oxidation state to every atom.
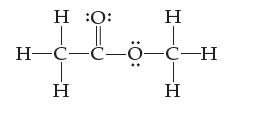
Transcribed Image Text:
Η :Ο: H¬C-C-0-C-H ά Η Η Η
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
a The problem with using the shortcut method to assign oxidation numbers to the atoms in methyl acet...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
The hydroquinone molecule can be converted to the quinone molecule as shown below: You may want to use a combination of shortcut rules and dot diagrams to assign oxidation numbers before answering...
-
Consider the three compounds shown below and then answer the questions that follow: a) Which two compounds are constitutional isomers? b) Which compound contains a nitrogen atom with trigonal...
-
F 6 I 2 O 4 is an interesting compound whose structure is shown below. Complete the dot diagram (add lone pairs) and then determine the oxidation state for every atom in the molecule not using the...
-
I currently have to complete an assignment on normalization anddenormalization, but I'm lost and don't know hat to do! General Instructions You must answer the following questions and create a...
-
Steward Inc. sells a product for $40 per unit. The variable cost is $30 per unit, and fixed costs are $15,000. Determine (a) The break-even point in sales units and (b) The breakeven point in sales...
-
A review of the ledger of D. J. Moore Company at December 31, 2012, produces the following data pertaining to the preparation of annual adjusting entries.1. Salaries and Wages Payable $0. There are...
-
A \(66-\mathrm{kg}\) person experiences a gravitational force of about \(660 \mathrm{~N}\). Yet, if this person were to jump onto a spring scale, the scale would briefly read about \(2400...
-
Tempe Office Services and Supplies (TOSS) provide various products and services in the Tempe Research Park, home to numerous high-tech and bio-tech companies. Making color copies is one of its most...
-
When calculating cash inflow or outflow, you must use the accrual accounts reported on the Income Statement, along with the related balance sheet accounts. What are the 2 balance sheet accounts that...
-
What do the terms oxidation and reduction mean with regard to valence electrons?
-
Consider ClO and AlCl 3 . For one of these substances, the halide shortcut rule works. For the other, it does not. (a) Which one does it work for, and why? (b) Why doesnt it work for the other? (c)...
-
Each extra worker produces an extra unit of out-put up to six workers. As more workers are added, no additional output is produced. Draw the total product of labor, average product of labor, and...
-
2. Consider the following program TotalCharge as an implementation of the core component of the Total Charge application: Program TotalCharge 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. begin...
-
Write a C++ console application that prints a solid rhombus based on inputs of character and number from the user. Use a validation loop to prompt for and get from the user a number (N) in the range...
-
Compare and contrast the various references that these artists make to the world beyond the scenes they depict? (city life, continuity with an actual chapel). Why do you think that Campin included...
-
GOVERNMENT 2 Case Study 2: Sally Sally is a 41-year-old woman with two school-age children. Sally has come to the Cultural Hub Community Centre, following a referral from her GP after breaking down...
-
How to calculate the expansion of a metal rod with a temperature increase. - Show work to get credit. How much will a 10-m-long copper rod at 20C expand when the temperature is changed from 0 to 100C?
-
In integrative problem 85 in Chapter 4, you were asked to calculate Carmins gross income for 2011. This is the second phase, which provides the additional information necessary for you to calculate...
-
Explain the differences and similarities between fringe benefits and salary as forms of compensation.
-
A popular circus act features daredevil motorcycle riders encased in the ???Globe of Death??? (Fig. P5.65), a spherical metal cage of diameter 16 ft.? (a) A rider of mass 65 kg on a 125-cc (95-kg)...
-
An ancient and deadly weapon, a sling consists of two braided cords, each about half an arm???s length long, attached to a leather pocket. The pocket is loaded with a projectile made of lead, carved...
-
A man stands 6.0 ft tall at sea level on the North Pole as shown in Figure P5.62.? (a) What is the difference in the value of g (the gravitational acceleration) between his head and his feet?? (b)...
-
2. Think of your own social support network. List all persons you have interacted with in the past month. Next, rank those persons on the list whom you perceive to be supportive in significant ways....
-
What are some of the significant similarities and differences among the field of counseling psychology and clinical psychology? What distinguishes counseling psychology from other fields of...
-
You want to buy a $ 6 5 0 , 0 0 0 house. If you plan to make a 2 0 % down payment, how much will you need to borrow? You plan to get a 1 5 - year mortgage. If the APR is 4 . 8 % , what will your...

Study smarter with the SolutionInn App


