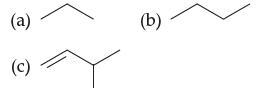
Convert each line drawing to a structural formula, then give the molecular formula and describe the hydrocarbon
Question:
Convert each line drawing to a structural formula, then give the molecular formula and describe the hydrocarbon as being linear or branched:
Transcribed Image Text:
(a) (c) (b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a Propane Lineangle formula a Butane Lineangle formula Linear c 3Methyl1but...View the full answer

Answered By

Surojit Das
I have vast knowledge in the field of Mathematics, Business Management and Marketing. Besides, I have been teaching on the topics Management leadership, Business Administration, Human Resource Management, Business Communication, Accounting, Auditing, Organizer Behaviours, Business Writing, Essay Writing, Copy Writing, Blog Writing since 2020. It is my personality to act quickly in any emergency situations when students need my services. I am very professional and serious in every questions students asked me at the time of dealing any projects. I have been serving detailed, quality, properly analysed research paper through the years.
4.80+
91+ Reviews
278+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Which molecule in Problem 17.39 is unsaturated? Data from Problem 17.39 Convert each line drawing to a structural formula, then give the molecular formula and describe the hydrocarbon as being linear...
-
Describe in words how you would do each of the following preparations. Then give the molecular equation for each preparation. a. CuCl2(s) from CuSO4(s) b. Ca(C2H3O2)2(s) from CaCO3(s) c. NaNO3(s)...
-
Describe in words how you would do each of the following preparations. Then give the molecular equation for each preparation. a. MgCl2(s) from MgCO3(s) b. NaNO3(s) from NaCl(s) c. Al(OH)3(s) from...
-
Which of the following statements are true about REST? Pick ONE OR MORE options Logical URLs should be used instead of physical URLS Adal URLs must always be used in REST response A paging technique...
-
A consumer is in equilibrium at point A in the accompanying figure. The price of good X is $5.a. What is the price of good Y?b. What is the consumer??s income?c. At point A, how many units of good X...
-
The units of an item available for sale during the year were as follows: There are 38 units of the item in the physical inventory at December 31. The periodic inventory system is used. Determine the...
-
Identify three stories from the past week that involve statistics in some way. In each case, write a brief statement describing the role of statistics in the story.
-
Determine the maturity date, interest in 2013 and 2014, and maturity value for a 90-day, 12 percent, $15,000 note from a customer dated December 1, 2013, assuming a December 31 year end. (Round to...
-
CASE (Pharma and Phenol) - Pharma has ownership interest in Phenol - Uses equity method - Rep on BOD is no longer there, they are accounting for it in a hybrid FV method (no longer equity method) -...
-
Hydrocarbons are often represented by line drawings. How is it possible to look at a line drawing and deduce the molecular formula of the compound?
-
What is the source of most hydrocarbons, and what ethical issues are involved in burning hydrocarbons as fuel?
-
Figure 5.15 shows the velocity, v, of an object (in meters/ sec). Estimate the total distance the object traveled between t = 0 and t = 6. v (m/sec) 40 30 20 10 1 2 3 4 5 Figure 5.15 6 1 (sec)
-
How does New Belgium introduce fun into the workplace? How does having fun keep employees engaged and enthusiastic about their work and the company? Kim Jordan and Jeff Lebesch, the husband and wife...
-
To avoid the NLRBs cease-and-desist order, what should Amalgamateds management have done differently in using problem-solving teams? Amalgamated Tool, a nonunion manufacturer of auto parts in...
-
The inflation target: Nearly every major central bank has chosen an inflation target of 2%. a. Why might a central bank choose a lower inflation target, for example, zero inflation? b. Why might a...
-
In 2003, Dinesh Thakur joined the Indian pharmaceutical company Ranbaxy. Thakur, who went on to become the companys director, uncovered fraudulent practices at the company in drug development,...
-
If push factors can be controlled by employers, what could stop them from doing so? Women leave the workforce at higher rates than men. In part, this may help explain why only about 2 percent of top...
-
What are the two primary reasons firms use standard process costing?
-
Comptech Ltd is a manufacturer of optical equipment. In September 2019, Ed Thompson the Chief Research Officer, attended a conference in Switzerland that focused on optical developments for the 21st...
-
For a system of atoms (in equilibrium) having two energy levels, show that at high temperatures where k B T >> E j - E i , the number densities of the two states tend to become equal.
-
Radiation at 21 cm pours down on the Earth from outer space. Its origin is great clouds of hydrogen gas. Taking the background temperature of space to be 3.0 K, determine the ratio of the transition...
-
With the Example 13.7 in mind, determine the average power per cubic meter radiated by the Nd:YAG laser rod, given that the transition occurs with an upper-level lifetime of 230 s.
-
In the Case: Great Western Hospital: High-risk Pregnancy Care What risk issues that are confronting the hospital.? What options they had to "manage" those risk issues ? And which of those options...
-
Direct versus Indirect Approach -choose which approach the Direct approach or the Indirect Approach is most appropriate to use for writing the messages listed below. Direct Approach or Indirect...
-
Based on what you learned in class and the guide posted in your classroom, choose the best communication channel to use to address each of the following communication situations. 5 points Oral...

Study smarter with the SolutionInn App


